Bioelectrical signals turn stem cells' progeny cancerous
Biologists at Tufts University School of Arts and Sciences have discovered that a change in membrane voltage in newly identified "instructor cells" can cause stem cells' descendants to trigger melanoma-like growth in pigment cells. The Tufts team also found that this metastatic transformation is due to changes in serotonin transport. The discovery could aid in the prevention and treatment of diseases like cancer and vitiligo as well as birth defects.
The research is reported in the October 19, 2010, issue of Disease Models and Mechanisms.
"Discovering this novel bioelectric signal and new cell type could be very important in efforts to understand the mechanisms that coordinate stem cell function within the host organism and prevent tumor growth. Ultimately it could enable us to guide cell behaviors toward regenerative medicine applications," said research leader and senior author Michael Levin, Ph.D., professor of biology and director of the Center for Regenerative and Developmental Biology at Tufts.
Co-authors on the paper were Tufts Postdoctoral Associate Douglas Blackiston, Research Associate Professor Dany S. Adams, Research Associate Joan M. Lemire and doctoral student Maria Lobikin.
Misregulation of stem cells is a known factor in cancers and birth defects. Recent studies have shown that stem cells exhibit unique electrophysiological profiles and that ionic currents controlled by ion channel proteins play important roles during stem cell differentiation. However, while many genetic and biochemical signaling pathways play a part in regulating the interplay between cells and the host organism, the role of bioelectric signals remains poorly understood, particularly when looking beyond artificial cultures to entire living organisms.
"One of the things we need to know is how cells know what to do in order to participate in the complex pattern of a host organism. The body normally tells cells 'don't become cancerous and go off on your own; instead, participate in keeping up the normal shape of all the tissues and organs, as individual cells age and die,'" said Levin.
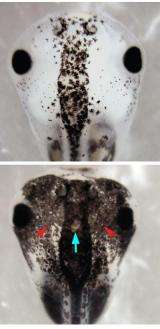
To determine how changes in membrane voltage regulate cell behavior in vivo, the Tufts researchers looked at a group of stem cells in Xenopus laevis frog embryos called the neural crest. Neural crest stem cells migrate throughout the body in vertebrates, including humans. They give rise to many cell types, including pigmentation cells called melancocytes, and contribute to structures such as the heart, face and skin. Congenital malformations of the neural crest are known to affect their descendent cells and cause birth defects.
The Tufts biologists manipulated the electrical properties of a special, sparse cell population present throughout the embryo by using the common anti-parasitic drug ivermectin to open the glycine gated chloride channel (GlyCl). The GlyCl channel is one of the many ion channels that control cellular membrane voltage and is a marker of this unique "instructor cell" population. Changing the chloride ion level to hyperpolarize or depolarize the cells in turn triggered abnormal growth in distant melanocytes derived from the neural crest. These pigment cells not only grew in greater numbers but also formed long, branch-like shapes and thoroughly invaded neural tissues, blood vessels and gut. This pattern is typical of metastasis.
The ability of these GlyCl-expressing cells to radically change the shape, position, and quantity of a different cell type (melanocytes) revealed a new and potentially highly important cell type -- an instructor capability that can change the behavior of other cells a considerable distance away.
The researchers achieved similar results when they used a variety of different methods to manipulate transmembrane potential. Therefore, they concluded that the impact was triggered by the voltage change itself and was not intrinsically dependant on ivermectin, chloride flow or the GlyCl channel.
Testing of human epidermal melanocytes in a depolarizing medium also showed a shape change similar to that found in the Xenopus tadpoles.
The researchers also addressed the question of how cells sensed depolarization and converted this biophysical signal into changes in distant cells' behavior. After testing three potential mechanisms, they found that transport of serotonin (a neurotransmitter that can be modulated to regulate mood, appetite and other functions) across the cell surface was the likely messenger.
The Tufts researchers note that analysis of other ion channels might reveal other instructor cells that can signal and change the behavior of various important cells in the body. Learning to identify and manipulate such cell types may reveal additional roles for ion flows and establish a new model for control of cell behavior in regenerative medicine and oncology.
Levin and his colleagues are already pursuing avenues for early, non-invasive cancer detection using voltage-sensitive dyes and exploring techniques to normalize cancer by repolarizing abnormal cells and instructor cells.
More information: "Transmembrane voltage gradient in GlyCl-expressing cell population controls behavior in neural crest derivatives in vivo," Douglas Blackiston, Dany S. Adams, Joan M. Lemire, Maria Lobikin and Michael Levin, Disease Models and Mechanisms, October 19, 2010.













