Final child vaccinated in clinical trial of new TB vaccine
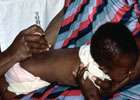
The final participant in a clinical trial to assess the safety and efficacy of a new vaccine against tuberculosis (TB) has been vaccinated.
The vaccination of the final child marks a significant milestone in the development of the MVA85A/AERAS-485 vaccine candidate, the most advanced of any of the new generation of preventative TB vaccines currently being investigated.
Tuberculosis kills 1.8 million people per year, and more than 2 billion people worldwide are infected with TB - approximately one in every three people on the planet. Bacille Calmette-Guérin (BCG) is currently the only available vaccine against the disease. It provides some protection against TB in children; however, it provides only variable protection against pulmonary TB, which accounts for most of the worldwide disease burden. New vaccines, therefore, are urgently needed to combat the problem.
The new vaccine, called MVA85A/AERAS-485, is given to children after the BCG and is designed to boost the body’s immune response to the vaccine, improving the level of protection. It was originally developed at the University of Oxford by Dr Helen McShane, a Wellcome Trust Senior Clinical Research Fellow, working with Dr Sarah Gilbert, a Reader in Vaccinology, and Professor Adrian Hill, a Wellcome Trust Principal Research Fellow.
A double-blinded phase IIb trial of MVA85A/AERAS-485 was launched in 2009 and conducted at the TB vaccine research site of the South African Tuberculosis Vaccine Initiative (SATVI) of the University of Cape Town, in partnership with Aeras, the Oxford-Emergent Tuberculosis Consortium Ltd and the Wellcome Trust.
Dr Michele Tameris, the Vaccine Trials Manager at the SATVI Worcester site, said: "Our team has worked tremendously hard to vaccinate our target of 2784 babies in just under two years. The trial is significant for being the most advanced of all TB vaccine candidates being tested in clinical trials, as well as the most babies enrolled in any TB vaccine trial. As a phase llb trial, it is designed to test for safety in a large group of babies as well as to see whether it works to prevent TB. It has already been successfully tested for safety in smaller groups of babies. Understandably, this final vaccination is a proud milestone for SATVI and its partners."
"We are pleased to report that the trial has run smoothly, with no vaccine-related serious adverse events being reported", added Dr Hassan Mahomed, who leads the trial as Principal Investigator and SATVI Co-Director.
"We are extremely proud of this achievement and are eager to see the study results, which are expected to be available in mid-2012," said Dr Helen McShane. "This milestone brings us a step closer to potentially having a new TB vaccine, from which millions of people around the world would benefit."
Following vaccination, babies are being followed up for around two years to see whether the new vaccine has provided protection against TB. The study is expected to run until the middle of 2012.
The trial has been part-funded by a Wellcome Trust Strategic Translation Award.















