Fetal programming of disease risk to next generation depends on parental gender
Overexposure to stress hormones in the womb can program the potential for adverse health effects in those children and the next generation, but effects vary depending on whether the mother or father transmits them, a new animal study suggests.
"This research sheds light on how babies who are exposed in the womb to excessive levels of stress hormones, known as glucocorticoids, can pass on the health effects to their own children, and how the effects vary between mothers and fathers," said the study's principal investigator, Amanda Drake, MD, PhD, a senior clinical fellow at the University of Edinburgh in Scotland.
Glucocorticoid levels may become raised during pregnancy if, for example, the mother experiences stress or illness or receives glucocorticoid drugs for treatment of illness or premature labor. Excess glucocorticoid exposure of the fetus can reduce birth weight and raise blood pressure later in life in animals and humans, and babies born with low birth weight are at increased risk of diabetes and heart disease in adulthood, Drake said.
"This has led to the concept of fetal programming, suggesting that the environment experienced in the womb can affect development, resulting in an increased risk of later disease. This increased disease risk can be passed to the next generation," Drake said.
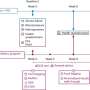
Using a rodent model of early life programming, Drake and colleagues studied the effects of glucocorticoid overexposure, with the drug dexamethasone, during the last week of gestation. They studied the effects on the directly exposed offspring and on their offspring. Their prior research showed that the low birth weight induced by prenatal exposure to dexamethasone transmits to a second generation through both male and female rats, according to Drake.
This new research showed that although birth weight is reduced in the offspring of male or female rats that were exposed to dexamethasone during fetal development, this effect was more pronounced in the offspring of male rats exposed to excess glucocorticoids during development in the womb.
Additionally, although birth weight was reduced in the second generation of rats, the genes that were affected differed from those seen in their parents, Drake said. In the first generation, glucocorticoid overexposure in the womb affected genes in the liver of the fetus and in the placenta. This increased the likelihood of the baby rats having a low birth weight and increased their risk of developing diabetes and heart disease in later life, she said.
However, the genes affected in the second generation depended on whether the mother or the father had been exposed to glucocorticoids while developing in the womb, the authors reported. These affected genes that could produce adverse health effects included genes important in growth and the transport of nutrients across the placenta.
Regarding the study, which was funded by the U.K. Medical Research Council, Drake said, "It could help inform future research to find interventions that could prevent diseases such as diabetes and high blood pressure," Drake said.