Researchers develop new tool in fight against apoptosis-related disease
(Medical Xpress) -- Though apoptosis is hardly a household word, it’s been estimated that more than half of all diseases for which we have no suitable treatment are related to malfunctions in apoptosis, also known as programmed cell death. It’s a body’s way of getting rid of mutated, damaged, old and other possibly harmful cells, and many researchers believe that mastering it could lead to a new cancer treatment, for example.
Now a chemist at the University of Massachusetts Amherst, Jeanne Hardy, with colleague Kimberly Tremblay of the veterinary and animal sciences department and members of their labs, have discovered a way to control an enzyme, one of a group known as caspases, that regulate apoptosis. Their work builds on Nobel Prize-winning chemistry studies by others using a visual marker that emits green light, Green Fluorescent Protein (GFP). It allows scientists to watch and film enzyme activities that control key processes inside a living organism.
"Caspases are like molecular scissors that cut certain key proteins in the cell, leading to apoptosis or cell suicide," Hardy explains. "We’ve made a ‘reporter’ named Caspase Activatible-Green Fluorescent Protein, which allows us to watch in real time when a cell starts to commit suicide and to track many molecular events including where the caspases are active and when. As far as we know, we’ve made the first GFP that can respond to an enzymatic action." Findings will appear in an upcoming issue of the Journal of Biological Chemistry.
In almost all cancers, cancer cells turn apoptosis down which leads to unregulated abnormal cancer cell division and tumor growth. Planned cell death is also seen in more normal cell function, for example during development in all animals, apoptosis stops growth of cells in certain areas such as in the limb buds of embryos, so properly shaped fingers or flippers will form in the correct place.
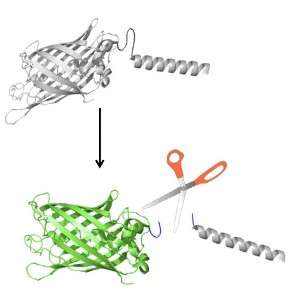
To modify the GFP, Hardy and colleagues appended onto its DNA a sequence or short tail that can be cut by the caspase and keeps the GFP "turned off" and temporarily dark until it is acted upon by a caspase enzyme. Developing a black version of GFP that only starts to emit the green fluorescence after apoptosis begins has allowed them to watch it turn on. Different brightness levels indicate the enzyme’s activity level.
A crystallographer and protein engineer who determines their structure and designs proteins to perform new functions, Hardy says the accomplishment of taking two different proteins and melding them together into an apoptosis reporter is crucial for observing cell death in real time. They make movies by taking a screen shot every 15 minutes over seven hours, then compressing the file so events unfold in time-lapse.
"For example, when some of the pores that protect the nucleus are cut by caspases, it allows Caspase Activatible-GFP to move into the nucleus. We have caught this part of the suicidal process on film. We have also studied the chemical reaction that allows Caspase Activatible-GFP go from the ‘turned off’ state to a bright fluorescent green ‘turned on’ state. We now understand the molecular details of this transformation," she says.
Now that Hardy, Tremblay and colleagues understand the chemistry of Caspase Activatible-GFP, they plan to make reporters that span the color spectrum. These new reporters are also capable of being turned on in various fluorescent colors in response to other molecular scissors involved in other important cellular and enzymatic processes, including watching where apoptosis happens during development to form body parts.
















