Unraveling the ins and outs of brain development
The embryonic nervous system is a hollow tube consisting of elongated neural progenitor cells, which extend from the inner to the outer surface of the tube. In a section inside the tube called the ventricular zone (VZ), these cells divide and produce immature neurons that migrate outwards. This involves well-characterized movements that are coupled to cell division. After a cell divides at the inner-most VZ region, the nuclei migrate to the outer region, where they synthesize new DNA before returning.
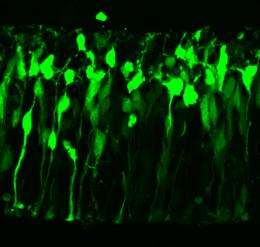
To determine how the direction of movement is coupled to the cell division cycle, Yoichi Kosodo and colleagues in Matsuzaki's group at RIKEN Center for Developmental Biology labeled nuclei in the embryonic mouse brain with green fluorescent protein. This enabled them to not only track their movements in cultured brain slices using a video-imaging system, but also correlate their positions with phases of the cell cycle. They found that outward nuclear migration involves back and forth ‘ratcheting’ motions and occurs more slowly than inward migration.
Importantly, they discovered that blocking the cell cycle before DNA synthesis caused nuclei to accumulate at the outer VZ surface (Fig.1), and reduced outward migration. Nuclei migrating back inwards normally crowd out those just finished dividing, thus pushing them away from the inner VZ surface.
Examining their results further, the researchers computationally modeled nuclear migration, and incorporated fluorescent magnetic beads into the inner VZ surface. They observed the beads moving away from the inner VZ surface, and remaining at its outer region.
The researchers also showed that inward migration is closely linked to microtubule reorganization orchestrated by a protein called Tpx2, which is initially expressed in the nuclei of progenitors before moving to the mitotic spindle. This separates newly duplicated chromosomes. Translocation of Tpx2 to the cell region nearest the inner VZ surface promotes migration of the nucleus in that direction by microtubule re-organization. Reducing Tpx2 activity lowered the velocity of inward migration, but introducing the human Tpx2 gene into the cells lacking Tpx2 restored normal speed.
The researchers conclude that two mechanisms maintain brain structure during development. One couples cell migration to the cell cycle, and occurs independently of other cells, with Tpx2 providing an active driving force; and the other involves interactions between the nuclei in the VZ.
More information: Kosodo, Y., et al. Regulation of interkinetic nuclear migration by cell cycle-coupled active and passive mechanisms in the developing brain. The EMBO Journal 30, 1690–1704 (2011).