Immunity restrained by ion influx
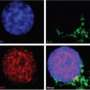
B cells maintain stockpiles of calcium ions (Ca2+), which are released during the course of the immune response. When the presence of a foreign antigen stimulates the B cell receptor (BCR) complex, these internal reserves of Ca2+ get released into the cell, subsequently triggering the opening of channels in the cell membrane that allow the entry of even more Ca2+.
Immunologists generally considered these ions as essential currency for many key cellular processes. “Ca2+ signaling in B cells is widely assumed to be responsible for functions including B cell development, immune response and antibody production,” says Yoshihiro Baba of the Immunology Frontier Research in Osaka University and formerly with the RIKEN Research Center for Allergy and Immunology in Yokohama. However, the direct effects of this bulk Ca2+ entry, also known as store-operated Ca2+ (SOC) influx, are poorly understood.
To examine the importance of this mechanism, Baba and colleagues genetically engineered mice whose B cells lack the genes encoding STIM1 or STIM2, two proteins involved with SOC influx. Their results suggest that this pathway plays a far more narrowly defined role than was previously expected.
The researchers determined that these two proteins cooperatively contribute to the management of Ca2+ influx, and facilitate B cell proliferation following BCR-mediated signaling. However, they appear to have no role in the actual immune response, as mice with STIM1- and STIM2-deficient B cells were still capable of mounting an antibody response against foreign antigens.
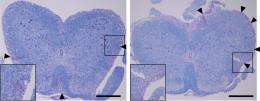
On the other hand, both factors proved important for the function of regulatory B cells, which produce anti-inflammatory factors such as interleukin-10 (IL-10) and help prevent the immune system from over-reacting or attacking host tissues. Without these STIM proteins, mouse B cells produced only minimal amounts of IL-10. Baba and colleagues determined that the absence of STIM1 and STIM2 greatly exacerbates the incidence and severity of inflammatory pathology in a mouse model of multiple sclerosis (Fig. 1). Since the action of IL-10 suppresses a variety of autoimmune conditions, these findings may indicate a general mechanism that could be targeted for the treatment of such disorders. “Our study suggests that a failure to balance Ca2+ levels may lead to autoimmune disease. This is a very exciting finding,” says Baba.
He and his colleagues are now keen to better understand the Ca2+-dependent anti-inflammatory B cells. “We are trying to show when and where regulatory B cells function, and what cells are targeted by them,” he says, “and to understand what type of inflammation—chronic or acute—is sensitive to IL-10-producing B cells.”
More information: Matsumoto, M., et al. The calcium sensors STIM1 and STIM2 control B cell regulatory function through interleukin-10 production. Immunity 34, 703–714 (2011).