November 7, 2011 report
Cell-replacement therapy could lead to Parkinson's treatment
(Medical Xpress) -- Researchers have developed an efficient way of making specific cells in high numbers that have worked in mice to reverse the damage caused by Parkinson’s disease and hope to be testing these cells in humans within the next few years.
While stem cell researchers have been able to produce the dopamine-producing neurons from human stem cells before, the stems cells didn’t seem to work as original cells would. The researchers discovered that subtle differences in the way these stem cells are grown can affect how they react when implanted.
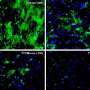
In 2009, Lorenz Studer from the Sloan Kettering Institute and his team created a new method for creating cells that is closer to the way they naturally form. These new cells have more of the characteristic molecular markers that are present in the naturally occurring dopamine-producing cells in the human brain.
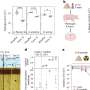
This new research, published in Nature, shows that the researchers have discovered a way to make these new cells more efficiently and in larger quantity.
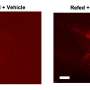
These cells have been tested in mice and rats with symptoms similar to Parkinson’s and within five months, the motor function in the rats had improved. Researchers have implanted the cells into rhesus monkeys, whose brain is closer to that of humans, but researchers say it is still too early to see the results.
The plan for Studer and his team now is to begin creating the cells on a larger scale in a U.S. Food and Drug Administration approved laboratory. Their goal is to create enough cells to graft 100 patients. It is believed by Studer that this will take about a year or so. Researchers plan to conduct extensive safety testing on the cells to make sure there is not a chance of the cells doing something unexpected after implantation.
More information: Dopamine neurons derived from human ES cells efficiently engraft in animal models of Parkinson’s disease, Nature (2011) doi:10.1038/nature10648
Abstract
Human pluripotent stem cells (PSCs) are a promising source of cells for applications in regenerative medicine. Directed differentiation of PSCs into specialized cells such as spinal motoneurons1 or midbrain dopamine (DA) neurons2 has been achieved. However, the effective use of PSCs for cell therapy has lagged behind. Whereas mouse PSC-derived DA neurons have shown efficacy in models of Parkinson’s disease3, 4, DA neurons from human PSCs generally show poor in vivo performance5. There are also considerable safety concerns for PSCs related to their potential for teratoma formation or neural overgrowth6, 7. Here we present a novel floor-plate-based strategy for the derivation of human DA neurons that efficiently engraft in vivo, suggesting that past failures were due to incomplete specification rather than a specific vulnerability of the cells. Midbrain floor-plate precursors are derived from PSCs 11 days after exposure to small molecule activators of sonic hedgehog (SHH) and canonical WNT signalling. Engraftable midbrain DA neurons are obtained by day 25 and can be maintained in vitro for several months. Extensive molecular profiling, biochemical and electrophysiological data define developmental progression and confirm identity of PSC-derived midbrain DA neurons. In vivo survival and function is demonstrated in Parkinson’s disease models using three host species. Long-term engraftment in 6-hydroxy-dopamine-lesioned mice and rats demonstrates robust survival of midbrain DA neurons derived from human embryonic stem (ES) cells, complete restoration of amphetamine-induced rotation behaviour and improvements in tests of forelimb use and akinesia. Finally, scalability is demonstrated by transplantation into parkinsonian monkeys. Excellent DA neuron survival, function and lack of neural overgrowth in the three animal models indicate promise for the development of cell-based therapies in Parkinson’s disease.
© 2011 Medical Xpress