Cancer drug improves memory in mouse model of Alzheimer's disease
A compound that previously progressed to Phase II clinical trials for cancer treatment slows neurological damage and improves brain function in an animal model of Alzheimer's disease, according to a study in the March 14 issue of The Journal of Neuroscience. The findings suggest the drug epothilone D (EpoD) may one day prove useful for treating people with early-stage Alzheimer's disease.
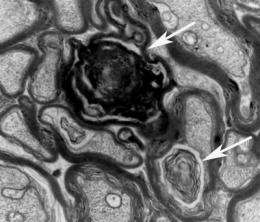
Nerve cells in people with Alzheimer's disease contain tangles — distorted clumps made up of the protein tau. Under normal circumstances, tau helps stabilize structures in nerve cells known as microtubules, which help transport nutrients and other substances from one end of the cell to the other. A growing body of evidence suggests the disruption in normal tau function during Alzheimer's disease could cause microtubules to break down, resulting in nerve cell damage.
In a previous study, Kurt Brunden, PhD, and colleagues at the University of Pennsylvania's Perelman School of Medicine and Department of Chemistry treated young mice that had been bred to develop symptoms similar to Alzheimer's disease later in life with EpoD. Treatment with EpoD — a drug that stabilizes microtubules — prevented microtubule breakdown and cognitive difficulties associated with Alzheimer's disease.
In the new study, Brunden's group examined the effects of EpoD on older mice that already displayed memory deficits and tau tangles similar to people with Alzheimer's disease. The researchers, led by first author Bin Zhang, PhD, administered EpoD weekly to these older Alzheimer's model mice for three months before evaluating them. Compared with Alzheimer's mice that did not receive the treatment, the EpoD-treated mice had reduced formation of tau tangles in the brain. These mice also outperformed animals that did not receive the drug in learning and memory tests.
"These results suggest that EpoD might have therapeutic benefit in Alzheimer's disease and related neurodegenerative diseases, such as frontotemporal lobar degeneration, where abnormal tau tangles are present," Brunden said.
EpoD uses a similar mechanism to stabilize microtubules as the FDA-approved cancer drug paclitaxel (Taxol™), Brunden explained. However, unlike Taxol™, EpoD efficiently crosses the blood-brain barrier — the network of blood vessels that keeps potentially harmful molecules from entering the brain. Because microtubule-stabilizing compounds are known to cause side effects at doses required for cancer treatment, Brunden's group administered a low dose of EpoD and closely tracked side effects. The mice displayed no side effects from the three-month EpoD treatment.
"EpoD readily enters the brain, where it appears to persist for a much longer time than in the blood. This may explain why low doses were both effective and safe in the mouse model of Alzheimer's disease," Brunden said.
Lester Binder, PhD, an expert in microtubule-associated proteins from Northwestern University Medical School who was not involved with the study, expressed optimism about the findings. However, he cautioned, "Although EpoD did not appear to cause problems in the mice at the doses given, it was administered for only three months, whereas a person would likely have to take the drug for years, as diseases featuring tau tangles are chronic."