Older patients with certain breast cancer subtype may not benefit from radiation therapy
Local breast radiation therapy may not be necessary for women with the luminal A subtype of breast cancer, particularly those aged older than 60, according to study results presented at the AACR Annual Meeting 2012, held here March 31 – April 4.
“Local breast radiation therapy, however, is still of benefit and is required for all the other breast cancer subtypes,” said Fei-Fei Liu, M.D., staff radiation oncologist at Princess Margaret Hospital, senior scientist at the Ontario Cancer Institute and professor at the University of Toronto, Ontario, Canada.
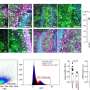
The researchers performed molecular subtyping for estrogen receptor (ER), progesterone receptor (PR), Ki-67, HER2, epidermal growth factor receptor and cytokeratin 5/6 on 304 tumor blocks from 769 women with breast cancer. These women had participated in a randomized trial in which they were assigned to tamoxifen and whole-breast radiation therapy or to tamoxifen alone. Based on the immunohistochemistry results, researchers classified patients into six categories: luminal A, luminal B, luminal-HER2, HER2-enriched, basal-like or triple-negative phenotype-nonbasal. They followed the patients for a median of 10 years.
Women in the luminal A subgroup, defined as ER-positive, PR-positive, HER2-negative and low Ki-67 (<14%), had the best outcome, with a 10-year risk for local relapse of 8 percent with tamoxifen alone vs. 4.6 percent with both tamoxifen and breast radiation therapy.
For luminal A patients aged older than 60, the local breast relapse rate was even lower at 4.3 percent with tamoxifen alone vs. 6 percent for tamoxifen plus breast radiation therapy, indicating that local breast radiation therapy did not contribute to the outcome of this group of patients, according to the researchers.
On the other hand, for other breast cancer subtypes, local breast radiation therapy was of definite benefit, according to Liu. For example, women with luminal B tumors had a recurrence rate of 16.1 percent with tamoxifen alone vs. 3.9 percent with tamoxifen and radiation therapy.
“If our data are validated with a larger number of patient tumor samples, we would recommend that Ki-67 be added to our current standard panel of ER, PR and HER2 testing for all patients with newly diagnosed breast cancer,” Liu said. “If the luminal A subtype is identified for lymph node-negative patients, especially for those 60 years old or older, then a discussion can be undertaken with these patients that if they take tamoxifen (or an equivalent medication) for their breast cancer, we might be able to avoid breast radiation therapy.
“This is yet another powerful example of ‘personalized cancer medicine.’ When this information is combined with well-conducted randomized clinical trials, significant advances can be made whereby we can truly start to tailor therapies, based on new molecular markers, which can be introduced into routine clinical practice.”