March 22, 2013 weblog
Nanotools for neuroscience and brain activity mapping
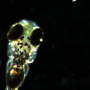
(Medical Xpress)—The ambitious and controversial Brain Activity Map (BAM), initiative instituted by a small group of researchers last year, has been steadily gaining momentum. Earlier this week, a proof-of-principle Zebrafish BAM was demonstrated with astounding clarity by a pair of researchers at the Howard Hughes Medical Institute.
Following on the heels of that work, an exhaustive 17-page compendium of current and soon-to-be brain mapping tools was published yesterday in ACS Nano by a rapidly snowballing list of disciples.
The BAM roster has been a carefully manicured player list from the beginning, and the role it has as ship wheel to this diffuse effort should not be underestimated. With the ranks now swelling to 27, each contributor to the paper has, in word or in spirit, contributed notably to the 185 referenced technologies on the paper. What we have here is not a research release, this is a textbook for the new neuroscience, and the journal choice, though not publicly accessible, hints at the desire to draw even more nanoscale researchers into the effort.
Media attention has channeled formative criticism to the effort in a way we have not seen before. Those sentiments on the cautionary take at least, might be summarized by likening the BAM scientists to cavemen having just discovered fire. Now sitting in the sand, they appear to be chartering a course to the internal combustion engine as they scribe on the ground with blunt bone instruments . The problem is that having just fleshed out how the brain's wiring, the connectome, might be extracted, the community elites just leapfrogged to the full activity map, or at least one for some of the lesser animals.
The most extravagant technology proposed is undoubtedly the DNA tickertape. It appears to have been developed initially, at least in part, by Northwestern University's Konrad Kording. Some of the earlier BAM papers show however that George Church, of human genome project fame, actually holds a patent that might cover some aspects of Kording's idea. In particular, Church seems responsible for the wickedly unique concept of engineering DNA polymerases to produce predictable errors that would in effect record conditions within the cell or device onto DNA tapes. Fortunately Church, having entered neuroscience some time ago, is also a BAM founding father. His "nucleic acid memory device" could be the means by which the spike activity of each neuron would be recorded.
Among the other wild exotica hinted at in the ACS Nano paper is the DNA barcode proposed by Anthony Zador, from the Cold Spring Harbor Lab. This device would use a genetically modified rabies virus to infiltrate the nervous system, and record every connection in the process, web-crawl style. While Zador is not an author on this or the previous BAM papers, his techniques would not only provide a way to deliver a connectome of a complex brain, they potentially could do it non-destructively. Furthermore, the barcode mechanism would perhaps be the ideal way to propagate the Kording-Church tickertape machinery from cell to cell, bundling topology and activity together.
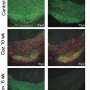
Many of the neurotools mentioned in the ACSNano paper are logical extensions of current technologies, just slightly smaller and a little higher in resolution. Recording cell activity with voltage-sensitive or calcium-imaging dyes, as was done in the Zebrafish map, may or may not be the process used ten years from now. Other ideas, like accessing neurons through fiber optic probes threaded through the vasculature to the capillaries, were re-invigorated, as were new sensors altogether like nanodiamond and nanogold devices.
Glaringly absent from this paper however, is a clear consensus of what exactly is to be done with these tools. The Zebrafish calcium map, for example, does not discriminate between neuron bodies, axons, dendrites, or synapses. The question of what level of detail is to be the goal of new studies still needs to be asked. This is a tough question because an activity map, like the connectome that would couch it, is rewritten on scales beneath our direct perception—not only is it a moving target, its trajectory is largely unknown. A long-term project such as this based in a set of technologies, as opposed to hypothesis-driven scientific inquiry, needs to balance fluidity with credibility.
Imagining what you would want to do if you were making a BAM of your own brain may emerge as the best way to set the project's goals. In that case, the researchers may not be going for the whole BAM right away—just the things they would want to know in enough detail to get some answers in the least destructive way possible. If they plow through a bunch of animal studies generating terabytes of data, but cannot then use those methods used to learn about our brains, they will not have been successful. Priority then is to be the nondestructive BAM, focused on those high-interest, highly accessible areas with the highest density of observables wherein the observation risks are low. How to do this is the question of the next BAM installment.
More information: Nanotools for Neuroscience and Brain Activity Mapping, ACS Nano, Article ASAP, DOI: 10.1021/nn4012847
Abstract
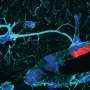
Neuroscience is at a crossroads. Great effort is being invested into deciphering specific neural interactions and circuits. At the same time, there exist few general theories or principles that explain brain function. We attribute this disparity, in part, to limitations in current methodologies. Traditional neurophysiological approaches record the activities of one neuron or a few neurons at a time. Neurochemical approaches focus on single neurotransmitters. Yet, there is an increasing realization that neural circuits operate at emergent levels, where the interactions between hundreds or thousands of neurons, utilizing multiple chemical transmitters, generate functional states. Brains function at the nanoscale, so tools to study brains must ultimately operate at this scale, as well. Nanoscience and nanotechnology are poised to provide a rich toolkit of novel methods to explore brain function by enabling simultaneous measurement and manipulation of activity of thousands or even millions of neurons. We and others refer to this goal as the Brain Activity Mapping Project. In this Nano Focus, we discuss how recent developments in nanoscale analysis tools and in the design and synthesis of nanomaterials have generated optical, electrical, and chemical methods that can readily be adapted for use in neuroscience. These approaches represent exciting areas of technical development and research. Moreover, unique opportunities exist for nanoscientists, nanotechnologists, and other physical scientists and engineers to contribute to tackling the challenging problems involved in understanding the fundamentals of brain function.
© 2013 Phys.org