March 20, 2013 report
Researchers image most of vertebrae brain at single cell level (w/ video)
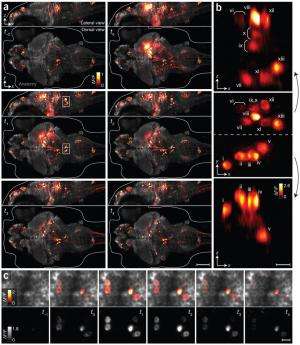
(Medical Xpress)—Misha Ahrens and Philipp Keller, researchers with the Howard Hughes Medical Institute have succeeded in making a near real-time video of most of a zebrafish's brain showing individual neuron cells firing. To create the video, as the team reports in their paper published in the journal Nature Methods, the two developed a type of modified light-sheet microscopy and used it in on genetically modified fish.
To create the video, the researchers turned to zebrafish in their larval state—their brains are transparent and small. To cause firing neurons to be visible they genetically altered the fish's brains, giving them a protein that glows when responding to changes in calcium ion levels, which happen when nerve cells fire. Next, they used a microscope that was able to broadcast a sheet of light through the fish's brain allowing for the detection of the firing neurons. The system recorded images every 1.3 seconds. The final step was stitching the images together to create a video. The result is nothing short of breathtaking—looking like something out of a science fiction movie's special effects department.
The video marks the first visual capture of most of a living vertebrae brain at the neuron level, as it works in near real-time and offers striking evidence of the complexity of the brain—even one as small as 100,000 neurons. The researchers say their video shows approximately 80 percent of the zebrafish's brain as it operates—though what all those firing neurons represent in particular, is still unknown.
The researchers are careful to point out that what they've accomplished does not portend the creation of a video of a human brain in action—our brains are much larger, have billions more neurons and perhaps more importantly, are not transparent and are covered by a thick skull. Instead they suggest that studying a simpler brain in action might help to explain how biological neural networks actually work, perhaps leading to theories that can be generalized over larger animals.
But before that can happen, the procedure the team has developed needs to be improved—neurons can fire at hundreds of times per second, which means a lot of firing in the video has been missed. Capturing at a faster rate would mean generating nearly unmanageable amounts of data—at the current rate, just one hour of capture creates a terabyte of data. Thus a new way to store and process the data must be developed.
More information: Whole-brain functional imaging at cellular resolution using light-sheet microscopy, Nature Methods (2013) doi:10.1038/nmeth.2434
Abstract
Brain function relies on communication between large populations of neurons across multiple brain areas, a full understanding of which would require knowledge of the time-varying activity of all neurons in the central nervous system. Here we use light-sheet microscopy to record activity, reported through the genetically encoded calcium indicator GCaMP5G, from the entire volume of the brain of the larval zebrafish in vivo at 0.8 Hz, capturing more than 80% of all neurons at single-cell resolution. Demonstrating how this technique can be used to reveal functionally defined circuits across the brain, we identify two populations of neurons with correlated activity patterns. One circuit consists of hindbrain neurons functionally coupled to spinal cord neuropil. The other consists of an anatomically symmetric population in the anterior hindbrain, with activity in the left and right halves oscillating in antiphase, on a timescale of 20 s, and coupled to equally slow oscillations in the inferior olive.
© 2013 Medical Xpress