Protein in fat cells that stimulates inflammatory signaling helps put gears in motion for onset of diet-induced obesity
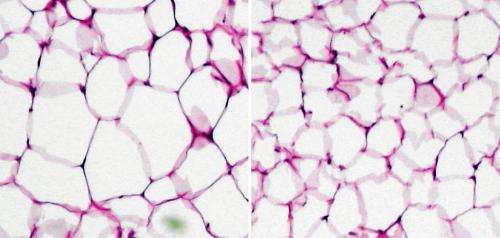
Poor diet and lifestyle choices set the stage for obesity and diabetes, but the immune system plays a relatively underappreciated role in accelerating this process. Metabolic changes in fat cells stimulate the release of inflammatory signals known as cytokines, which block insulin signaling at a cellular level, as well as other factors that recruit immune cells into fatty tissue to perpetuate the cycle of declining metabolic function.
Yoshio Hirabayashi, Yeon-Jeong Kim and colleagues from the RIKEN Brain Science Institute's Laboratory for Molecular Membrane Neuroscience have now begun to characterize the mammalian counterpart of a protein related to fat metabolism in fruit flies. Their latest work reveals that this mammalian protein, GPRC5B, is a key promoter of cytokine signalling.
Several years ago, Hirabayashi's team identified that the protein BOSS regulates glucose and fat metabolism in fruit flies2. In investigating the mammalian counterpart GPRC5B, they determined that this protein is strongly expressed in adipose tissue in mice, and contributes strongly to fat accumulation. After being fed a high-fat diet, mice genetically deficient in GPRC5B gained less weight and showed reduced body fat compared with wild-type mice, and generally exhibited a higher metabolic rate.
The researchers found that GPRC5B primarily localizes within cell membrane-based structures called 'lipid rafts', where it interacts with the signaling protein Fyn. Subsequent experiments revealed that the two proteins collaborate closely: Fyn helps to switch on GPRC5B, which in turn enables GPRC5B to strongly stimulate Fyn activity. "Fyn is usually activated by growth factors—including insulin—and cellular stress, which is crucially important for obesity and the inflammatory process in adipose tissue," says Hirabayashi.
The researchers view GPRC5B as a potentially important player in fueling the Fyn-mediated inflammatory response in diet-induced obesity, as evidenced by the reduced pro-inflammatory signaling in the fatty tissue of GPRC5B-deficient animals. A cellular signaling cascade known as the NF-κB pathway is critical in driving production of these cytokines, and Fyn-inactivating mutations sharply reduced NF-κB activation in cultured mouse fat cells, confirming the importance of GPRC5B-Fyn crosstalk. "GPRC5B may contribute to enhanced activation of localized Fyn," says Hirabayashi, "leading to the promotion of inflammatory signaling."
The research team now hopes to fill in the blanks for this signaling pathway. This includes identifying mechanisms that regulate GPRC5B activation and the mechanism by which GPRC5B and Fyn stimulate NF-κB activity to generate an inflammatory response—findings that should help illuminate how physiological stress associated with initial onset of obesity fuels subsequent disruption of metabolic regulation.
More information: Kim, Y. et al. GPRC5B activates obesity-associated inflammatory signaling in adipocytes. Science Signaling 5, ra85 (2012). dx.doi.org/10.1126/scisignal.2003149
Kohyama-Koganeya, A. et al. Drosophila orphan G protein-coupled receptor BOSS functions as a glucose-responding receptor: Loss of boss causes abnormal energy metabolism. Proceedings of the National Academy of Sciences USA 105, 15328–15333 (2008). dx.doi.org/10.1073/pnas.0807833105