Dopamine-producing neurons derived from bone marrow stem cells yield improvements in monkeys with Parkinson's disease
Parkinson's disease is a neurodegenerative disorder characterized by the death of dopamine-producing neurons in the midbrain, resulting in motor symptoms such as tremors and stiffness. The cause of cell death remains unknown and researchers have long sought a way to replace the lost dopamine-producing cells. A study led by Takuya Hayashi from the RIKEN Center for Molecular Imaging Science now suggests that in monkeys such neurons can be derived from bone marrow stem cells and then transplanted back into the brain to reverse the symptoms of this devastating disease.
Hayashi, Mari Dezawa from Tohoku University and their colleagues injected ten adult male cynomolgus monkeys (crab-eating macaques) with a neurotoxin that induces a Parkinson's-like condition. They then obtained bone marrow samples from the monkeys, isolated the marrow's mesenchymal stem cells (MSCs), and treated the cells with growth factors to direct them to differentiate into A9 dopaminergic neurons—the neuronal subtype that is most severely damaged in Parkinson's patients. The researchers subsequently transplanted the differentiated cells back into the forebrain of five of the donor monkeys, while the other five animals received a sham operation.
The procedure is an example of an 'autologous' transplantation, involving cells derived from and transferred to the same individual. Autologous transplantation eliminates the possibility of immune rejection, making this approach attractive for eventual use in the clinic.
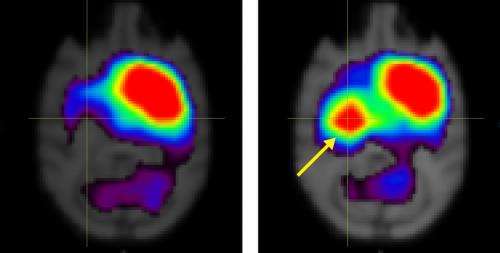
Several months later, the treated monkeys, but none of the untreated subjects, exhibited improvements in motor behaviors, such as performance in a hand-reach task. Positron emission tomography scans of the cell-engrafted monkeys' brains revealed a dramatic increase in the expression of dopamine transporter (DAT), a membrane-spanning protein that helps clear dopamine from the synapse (Fig. 1).
DAT expression in monkeys that received the transplanted neurons remained above pre-treatment baseline levels for more than seven months after the operation. Further analyses at nine months demonstrated the existence of cells positive for DAT and other markers indicative of dopaminergic neuron function in the engrafted striatum—the forebrain region in which the MSC-derived neurons were implanted. The findings are consistent with functional integration and survival of the transplanted tissue.
Hayashi next plans to compare the efficacy of transplanting differentiated versus native MSCs in this monkey model. He also hopes to start translating his system for human applications. "Our newly developed system of cell-based therapy restored motor function of animal models with Parkinson's disease," says Hayashi. "We should now test whether we can derive functional and viable dopaminergic cells from human MSCs."
More information: Hayashi, T. et al. Autologous mesenchymal stem cell-derived dopaminergic neurons function in parkinsonian macaques. The Journal of Clinical Investigation 123, 272–284 (2012). dx.doi.org/10.1172/JCI62516