May 24, 2013 report
Engineered cytomegalovirus protects monkeys from HIV equivalent
(Medical Xpress)—A new study by researchers in the US has shown that an ancient virus can be modified to help in the fight against the simian immunodeficiency virus SIV, which is the equivalent in monkeys to HIV in humans.
The research team, led by Scott G. Hansen of the Oregon National Primate Research Center and the Vaccine and Gene Therapy Institute, earlier (2011) reported on their research in modified rhesus cytomegalovirus (CMV) strains. These strains belong to a family of viruses that has evolved alongside their mammalian hosts and which affect the T cell immune response.
That study showed how the genetically engineered virus expressed simian immunodeficiency virus (SIV) proteins called RhCMV vectors. These vectors then allowed rhesus macaques (Macaca mulatta) vaccinated with the RhCMV to control SIV infection by modifying the T cell responses. The team reported that half of the rhesus macaques they vaccinated with the RhCMV/SIV vector were able to control SIV infection when challenged with a highly pathogenic strain.
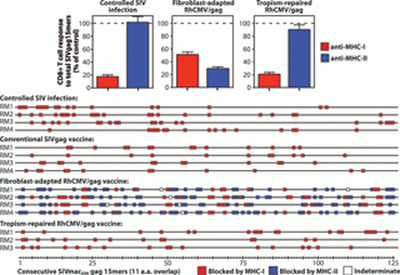
In the current study, the team describes their latest findings, which are that a certain modified strain of CMV (68-1 RhCMV) can provoke some immune response cells (CD8+ type T cells) to respond to more antigens than they usually do and produce a much more robust response. Immunodeficiency viruses are usually able to escape the normal responses of CD8+ T cells.
T cells, or T lymphocytes, represent a type of white blood cell that matures in the thymus and plays an important role in the immune system. T cells express either CD8 or CD4 glycoproteins on their surface. Those expressing CD4 glycoprotein are known as T helper cells, since they assist other blood cells involved in the immune response, while those expressing CD8 are known as cytotoxic T cells, since they destroy infected and tumorous cells.
T cells detect a small fraction of peptides derived from pathogens that are transported to the surface of cells by class I and II major histocompatibility complex (MHC) molecules. The CD4+ T helper cells recognize antigens transported by MHC-II, while the CD8+ cytotoxic T cells recognize antigens associated with MHC-I.
When 68-1 RhCMV is present, CD8+ T cells can detect SIV epitropes, which are fragments of antigenic proteins they normally miss. They can also respond to antigens transported by MHC-II, which are normally responded to only by CD4+ T cells.
The results show that CMV can be genetically engineered to affect the body's arsenal of CD8+ T cells so that they can recognize and mount an effective response against a wider range of peptides, and hence SIV epitopes.
The authors say their results may lead to advances in treatment of HIV in humans. The paper is published in today's edition of the journal Science.
More information: Cytomegalovirus Vectors Violate CD8+ T Cell Epitope Recognition Paradigms, Science 24 May 2013: Vol. 340 no. 6135 DOI: 10.1126/science.1237874
Abstract
CD8+ T cell responses focus on a small fraction of pathogen- or vaccine-encoded peptides, and for some pathogens, these restricted recognition hierarchies limit the effectiveness of antipathogen immunity. We found that simian immunodeficiency virus (SIV) protein–expressing rhesus cytomegalovirus (RhCMV) vectors elicit SIV-specific CD8+ T cells that recognize unusual, diverse, and highly promiscuous epitopes, including dominant responses to epitopes restricted by class II major histocompatibility complex (MHC) molecules. Induction of canonical SIV epitope–specific CD8+ T cell responses is suppressed by the RhCMV-encoded Rh189 gene (corresponding to human CMV US11), and the promiscuous MHC class I– and class II–restricted CD8+ T cell responses occur only in the absence of the Rh157.5, Rh157.4, and Rh157.6 (human CMV UL128, UL130, and UL131) genes. Thus, CMV vectors can be genetically programmed to achieve distinct patterns of CD8+ T cell epitope recognition.
© 2013 Medical Xpress