May 29, 2013 report
Controlling ventricular volume through ciliary beat frequency
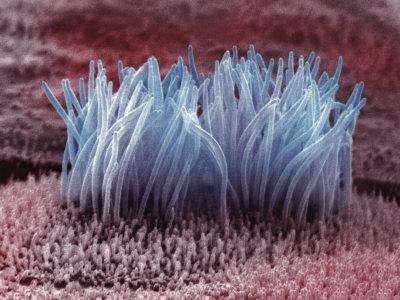
(Medical Xpress)—Motile cilia are present on the surface of many types of cells. When they are not working properly, respiratory problems, various sensory deficits, and occasionally, even a reversal of placement of the internal organs can occur. In the lining of the ventricles of the brain, cilia on the surface of ependymal cells facilitate mixing and circulation of cerebral spinal fluid (CSF). When these cilia fail to perform, narrow connecting aqueducts in the ventricular system can pinch off leading to uncontrollable swelling of the brain and head. A group of French researchers have recently discovered a critical mechanism in the regulation of beat frequency of the cilia on these cells. In their new paper in Nature Neuroscience, they show that melanin-concentrating hormone (MCH) is the molecular agent which steps up the beat cycle. They additionally demonstrate that transgenic mice lacking the receptor (MCHR)for this hormone have significantly increased ventricular volume, and altered flow of CSF. Previous studies have suggested that some forms obesity might result from an underlying ciliopathy, with MCH acting as a key control on appetite. These new results raise a cautionary note to the potential use of MCHR antagonists for therapeutic treatment.
When the French researchers initially looked at coronal (front-viewed) sections of the MCHR knockout mice, they found MCH-rich fibers in the ventral part of the third ventricle. These fibers presumably originate in specific parts of the lateral hypothalamus, an area also involved in regulation of appetite. Triple immunolabeling for glu-tubulin in cilia, and for vimentin in and MCHR in ependymal cells, established that only cells expressing MCHR were contacted by MCH fibers. These observations suggest that a dedicated control circuit may be in operation here rather than just a diffuse delivery of MCH through via the CSF.
The researchers developed a high-speed, bright-field videomicroscopy rig to test whether MCH directly regulated beat frequency. To validate their setup, they first showed that both serotonin and ATP, neuromodulators already known to increase beat frequency, had the desired effect in the third ventricle. They then found that MCH also increased the beat frequency by up to 40% in a concentration dependent fashion. For comparison, the spontaneous frequency for knockout MCHR mice was around 5 hz.
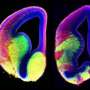
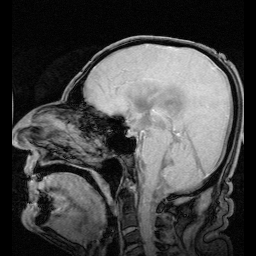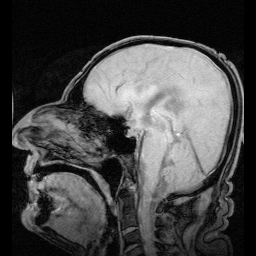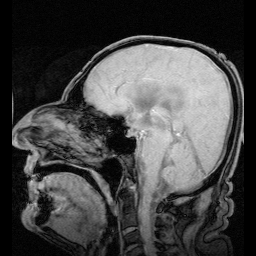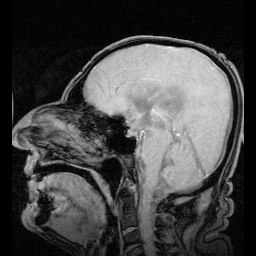
A decrease in beat frequency has been previously linked to ventricular enlargement, presumably through fluid accumulation. The researchers took an MRI scan of the MCHR mice and found increased volume in the lateral and third ventricles. These finding are consistent with the idea of reduced drainage of CSF through the central aqueduct.
The role of cilia in ventricular hydraulics is still being worked out. Undoubtedly the prime mover is the heart, as its pressure pulse penetrates through to the ventricles directly. The cilia likely contribute significant pumping action in the narrower channels, and also in small or developing brains. As they are generally just 15 microns or so in length, the primary effect of cilia may be in local mixing and distribution of CSF. The authors also suggest an auxiliary role might be to sense glucose, or otherwise meet energetic needs by redistributing metabolites.
To get a better idea of what might be going on inside ependymal cells, the researchers performed electrical recordings during application of MCH. They found that MCH hyperpolarized the cells by activating a calcium-dependent potassium channel. Stimulation of the lateral hypothalamus directly was found to increase ciliary beat frequency on the corresponding side. Blocking MCHR's abolished most of this effect, suggesting most of the response could be attributed to MSH.
Many cells in the brain, not just those lining the ventricles, maintain their cilia beyond the brief developmental period where they serve sensory or mechanical function in migration. Better understanding of the sometimes unanticipated functions of cilia will lead to a better understanding of neurons, and therefore brains in general.
More information: Melanin-concentrating hormone regulates beat frequency of ependymal cilia and ventricular volume, Nature Neuroscience (2013) doi:10.1038/nn.3401
Abstract
Ependymal cell cilia help move cerebrospinal fluid through the cerebral ventricles, but the regulation of their beat frequency remains unclear. Using in vitro, high-speed video microscopy and in vivo magnetic resonance imaging in mice, we found that the metabolic peptide melanin-concentrating hormone (MCH) positively controlled cilia beat frequency, specifically in the ventral third ventricle, whereas a lack of MCH receptor provoked a ventricular size increase.
© 2013 Medical Xpress