July 16, 2013 report
Cranial irradiation causes brain degeneration
(Medical Xpress)—Cranial irradiation saves the lives of brain cancer patients. It slows cancer progression and increases survival rates. Unfortunately, patients who undergo cranial irradiation often develop problems with cognitive functioning. To determine how radiation affects cognition, Vipan Parihar and Charles Limoli of the University of California, Irvine studied cranial irradiation in mice. They found that exposure to radiation causes degenerative changes to brain architecture similar to those observed in people with neurodegenerative conditions such as Alzheimer's disease and Huntington's disease. Their research appears in the Proceedings of the National Academy of Sciences.
Radiation therapy is the routine frontline treatment for almost all forms of pediatric and adult brain cancer because of its ability to forestall tumor growth. While it increases the lifespans of people diagnosed with brain cancer, cranial irradiation can reduce quality of life by causing irreversible cognitive impairment. Central nervous system (CNS) exposure to radiation causes problems with memory, learning, attention, processing speed and executive function.
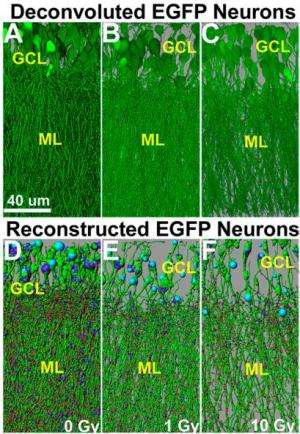
To understand how radiation exposure reduces cognitive ability, Parihar and Limoli exposed mice to either 1 or 10 Gy of radiation, doses much lower than the maximum dose the CNS can withstand before tissue damage occurs. After 10 or 30 days, the researchers killed the mice and dissected their brains. They then examined the hippocampus, which is associated with learning and memory.
Parihar and Limoli observed dose-dependent reductions in the area, length and branching of dendrites, projections on neurons that send and receive signals to and from other neurons. These reductions persisted after 30 days. The number and density of dendritic spines, bulbous extensions on dendrites, also decreased. Dendritic spines regulate CNS connectivity, are associated with memory storage and play an important role in mediating brain plasticity. There is a positive correlation between number of dendritic spines and synaptic density, which in turn correlates with cognitive ability. The researchers also identified significant changes in levels of pre and post-synaptic proteins.
Reduced dendritic complexity is a characteristic of Alzheimer's disease, Huntington's disease, recurrent depressive illness and epilepsy. Dendritic spine abnormalities are associated with Huntington's disease, temporal lobe epilepsy, AIDS-related dementia, Down syndrome, Rett syndrome and Fragile-X syndrome.
Parihar and Limoli state that the reduction in dendritic spine density and the persistence of degenerative changes after one month is consistent with the irreversible reduction in cognitive functioning experienced by brain cancer survivors who have had cranial radiotherapy.
More information: Cranial irradiation compromises neuronal architecture in the hippocampus, PNAS, Published online before print July 15, 2013, doi: 10.1073/pnas.1307301110
Abstract
Cranial irradiation is used routinely for the treatment of nearly all brain tumors, but may lead to progressive and debilitating impairments of cognitive function. Changes in synaptic plasticity underlie many neurodegenerative conditions that correlate to specific structural alterations in neurons that are believed to be morphologic determinants of learning and memory. To determine whether changes in dendritic architecture might underlie the neurocognitive sequelae found after irradiation, we investigated the impact of cranial irradiation (1 and 10 Gy) on a range of micromorphometric parameters in mice 10 and 30 d following exposure. Our data revealed significant reductions in dendritic complexity, where dendritic branching, length, and area were routinely reduced (>50%) in a dose-dependent manner. At these same doses and times we found significant reductions in the number (20–35%) and density (40–70%) of dendritic spines on hippocampal neurons of the dentate gyrus. Interestingly, immature filopodia showed the greatest sensitivity to irradiation compared with more mature spine morphologies, with reductions of 43% and 73% found 30 d after 1 and 10 Gy, respectively. Analysis of granule-cell neurons spanning the subfields of the dentate gyrus revealed significant reductions in synaptophysin expression at presynaptic sites in the dentate hilus, and significant increases in postsynaptic density protein (PSD-95) were found along dendrites in the granule cell and molecular layers. These findings are unique in demonstrating dose-responsive changes in dendritic complexity, synaptic protein levels, spine density and morphology, alterations induced in hippocampal neurons by irradiation that persist for at least 1 mo, and that resemble similar types of changes found in many neurodegenerative conditions.
© 2013 Medical Xpress















