Bacterial toxin sets the course for infection
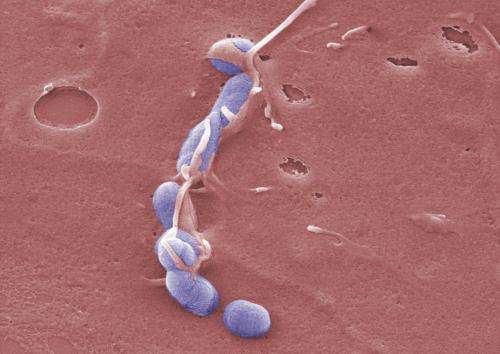
Braunschweig have now discovered what makes a specific strain of Yersinia pseudotuberculosis—one of the main instigators for these infections–so dangerous: the bacteria produce a molecule called CNFy that facilitates the infection process for them. It changes the host cells in a manner that enables the injection apparatus of Yersinia, which injects toxins into the cells, to work more efficiently. This strengthens the infection and leads to inflammation of the tissue.
Whether an immune cell divides, alarms other immune cells or dies is strictly controlled in our immune system. "Molecular switches" influence these processes and basically set the course for different pathways. In light of the evolutionary competition between the immune system and the microbes, researchers have found that bacteria produce different substances to manipulate the position of the switches to their advantage.
Scientists at the Helmholtz Centre for Infection Research (HZI) in Braunschweig and at the Hannover Medical School (MHH) have examined one of those substances in more detail. Their results have been published in the American scientific magazine PLOS Pathogens. The team led by Prof Petra Dersch, head of the Department "Molecular Infection Biology", became aware of the molecule called CNFy, because the bacterium Yersinia pseudotuberculosis produces it in large quantities.
Yersinia pseudotuberculosis is transmitted via contaminated food and can generate gastro-intestinal diseases. However, not all strains produce CNFy. The scientists had therefore assumed that it played no significant role. The interdisciplinary team has now shown that this was a mistake. "Bacteria only produce molecules that are useful for their purposes. Therefore, we wanted to know why Yersinia would need CNFy," Dersch says.
In order to elucidate its function, the scientists genetically modified a bacterial strain that usually forms CNFy in such a way that it lost the ability to produce this factor. "The altered bacterium was no longer capable of escaping the immune system of the host organism and could not cause disease," reports Janina Schweer, PhD student at the HZI. This is remarkable since the bacteria certainly have other pathogenic characteristics in their repertoire. They have large molecular complexes at their disposal with which they can inject destructive substances into the host cell. This is a very effective method to promote an infection. "It seems that this mechanism is not sufficiently active in some Yersinia. Apparently, the examined Yersinia strain needs CNFy so that its "molecular syringes" can inject sufficient quantities of active substances into the immune cells," explains Prof Jochen Hühn, head of the Department "Experimental Immunology" at the HZI. These active substances, mostly cell toxins, damage the immune cells. Many of the substances cause cell mortality. This facilitates the expansion of Yersinia within the infected organism. During advanced infections, inflammation occurs, as well as damage to the tissue.
The researchers have also identified the molecular target that CNFy manipulates, generating the dramatic consequences: this involves the so-called small Rho GTPases. These enzymes initiate a whole cascade of events, for example alteration of the cytoskeleton. This leads to pores in the host cell surface, through which bacterial syringes can more efficiently transport active agents into the cell. The observed cell mortality of the immune cells is introduced through Rho GTPases as well.
"We here have discovered a very clever strategy of Yersinia pseudotuberculosis. With the aid of CNFy, the bacterium manipulates the host cell in such a manner that the injection apparatus can work more effectively," explains Dersch. "It sets the course for an efficient infection and triggers onset of the disease."
The present study shows that CNFy is very important for Yersinia. At the same time, it emphasizes the central role of the injection apparatus that is deployed in a more robust manner via CNFy – it is, and remains, an important drug target for intervention measures.
More information: The cytotoxic necrotizing factor of Yersinia pseudotuberculosis (CNFy) enhances inflammation and Yop delivery during infection by activation of Rho GTPases. PLOS Pathogens, 2013.