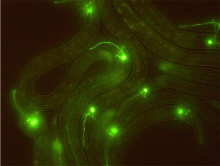
C. elegans has sensory neurons that detect environmental conditions
In order to survive, animals must be able to sense what is happening in the environment. Some animals have excellent sight, others a great sense of smell or taste. The senses are used to find food, mates, to avoid toxins and predators. The small worm C. elegans has a set of sensory neurons that detect various conditions in its environment, almost like a sense of smell. It is an ideal organism for studying how animals interact with their environment because its sensing systems and reactions are simple and can be understood at a genetic level. Members of OIST's Information Processing Biology Unit, led by Prof. Ichiro Maruyama, have recently uncovered the sensor for alkaline environments, or high pH, in C. elegans.
The paper was chosen as a Plenary Article by the editors of Neuroscience Letters because of, "especially meritorious work," according to the Editor-in-Chief, Dr. Stephen Waxman. Dr. Waxman says editors recommended the article because "outside experts called the work an interesting set of experiments that provides insights into how high pH is detected as a repellent stimulus by the sensory system."
pH is an important condition to sense because a pH too high or too low can cause harm to an organism. A low pH indicates acidity, and a high pH indicates alkalinity. Substances either too low or too high in pH can, in extreme cases, cause extensive tissue damage or death. Even small fluctuations can cause harm. In humans, a difference of only 0.2 pH units in the blood can lead to extreme health problems. Normally, the body's ability to sense pH prevents a serious problem from arising, but if these sensors are not functioning properly, or the body does not react appropriately, the results could be catastrophic.
In the tiny worm C. elegans, the researchers have taken advantage of the simple way the worm can sense alkalinity, or high pH, in its environment. In a previous paper, the group showed that worms prefer a slightly alkaline environment. Here, they demonstrate that the worms are averse to a very high pH by showing that the worms avoided areas of the experimental environment where the researchers created highly alkaline conditions. The researchers also identified the specific neuron responsible for detecting high pH. They did this by showing that when a specific sensing neuron, called the ASH neuron, was destroyed by laser microsurgery, the worms no longer avoided high pH. Going another step further, the researchers also identified some of the proteins involved in the response to high alkalinity, which make up a calcium channel. Using one of the great advantages of C. elegans, they were able to visualize the influx of calcium that occurred in a neuron in response to stimulus in a living worm. When the two proteins that form the calcium channels were mutated, the neuron was no longer activated.
The next goal for the researchers is to identify other key players involved in the response to alkaline environments. Toshihiro Sassa and Takashi Murayama, the first and second authors of the paper, respectively, say, "we would like to identify the other genes involved in downstream signaling from this calcium channel to understand how the signal leads to aversion to high pH." Sensing alkaline environments is only the first step; the worm must then quickly react to the stimulus by moving away. The researchers would like to understand the genes and neuronal networks involved in regulating the avoidance behavior that occurs after high pH is sensed. The ability to study such complex neurological responses in a simple animal like the worm helps scientists understand the more complex systems that exist in humans. It seems like Friedrich Nietzsche had it right when he said, "You have made your way from worm to man, and much in you is still worm."
More information: Toshihiro Sassa, Takashi Murayama, Ichi N. Maruyama, "Strongly alkaline pH avoidance mediated by ASH sensory neurons in C. elegans," Neuroscience Letters, Volume 555, 25 October 2013, Pages 248–252, dx.doi.org/10.1016/j.neulet.2013.06.001, www.sciencedirect.com/science/ … ii/S0304394013005338