Oligomers' role in the development of Parkinson's disease
Researchers at Aarhus University, Denmark, have drawn up the most detailed 'image of the enemy' to date of one of the body's most important players in the development of Parkinson's disease. This provides much greater understanding of the battle taking place when the disease occurs – knowledge that is necessary if we are to understand and treat Parkinsonism. However, it also raises an existential question because part of the conclusion is that we do not live forever!
Parkinson's disease is one of the most common neurological disorders, with about 7000 people suffering from the disease in Denmark alone. There is no cure, and the symptoms continue to get worse. The disease occurs because different nerves in the brain die. These include the nerve cells that form dopamine, which is known as the brain's 'reward substance' and which also helps control our fine motor skills.
A group of researchers from Aarhus University, the University of Southern Denmark (SDU) and the University of Cambridge has just published two studies in the prestigious Journal of the American Chemical Society (JACS) and Angewandte Chemie. These studies provide the best insight to date into the behaviour of a particular protein state that plays an important role in Parkinson's disease. In other words, they have created a detailed image of what is presumed to be the arch enemy we are up against in our understanding of Parkinsonism. It is an advanced antagonist, and one that functions with a considerable degree of unpredictability. "Fighting the enemy is by no means a Sunday outing," say the main authors of the results – Professor Daniel Otzen, Aarhus University, and his colleagues Nikolai Lorenzen and Wojciech Paslawski, who recently defended their PhD dissertations on this subject at Aarhus University's Interdisciplinary Nanoscience Centre (iNANO).
Protein aggregation kills nerve cells
Knowledge about what actually takes place in the brain when Parkinson's disease occurs and develops is absolutely necessary, not only for the prevention and treatment of symptoms, but also for possibly developing a cure one day.
However, getting to know the enemy is no easy task when it comes to understanding Parkinson's disease. The more we find out, the more complex the image becomes.
We already know that the disease – as well as other neurological disorders – arises because some protein structures in the body start clumping together. They stack themselves on top of each other and gradually form what are known as fibrils – long, thin needle-shaped structures. From a biochemical point of view, this is quite a boring process because the protein structures simply pile themselves on top of each other and – in principle – can continue to do so forever.
It is far more interesting to look at the intermediate stages leading up to the aggregation. It turns out here that when the proteins form fibrils, a kind of intermediate aggregation process also takes place to form oligomers, which consist of a small number of protein molecules that clump together. It is presumably the oligomers that kill the nerve cells and cause the symptoms of Parkinson's disease. So oligomers are the enemy we want to control.
Ground-breaking new knowledge about the enemy
In their two studies, the researchers from Aarhus University, the University of Southern Denmark and the University of Cambridge provide the most well-documented description of oligomers to date. Until now, the general perception was that oligomers were precursors of the fibrils. As it turns out, however, it is rather the antagonists or competitors of the fibrils that are the precursors, and these are capable of slowing down the formation of fibrils.
The researchers discovered that there are different kinds of oligomers. If we look at the size, there are two types that are quite intimately connected. A somewhat small oligomer that is very well defined and a larger one that is virtually a chain composed of the smaller ones. The oligomers are thus capable of linking up in the same way as the fibrils, but in chains that inhibit the fibrils. "You could say that they put a spanner in the works regarding the formation of fibrils," says Professor Otzen. It could possibly be a help to shift the focus even further away from the fibrils, which are formed in a different process than the oligomers, and which should not be a target for pharmaceutical products on their own.
Description of the enemy
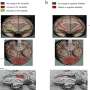
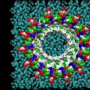
So what does our adversary look like? The researchers can now help provide us with a better answer. An oligomer consists of a very stable interior and a more diffuse sphere surrounding it, where the protein is not as compact and where it flaps around a little
And yet the image is even more complicated than that! Because if you take a closer look at the little oligomer, there are also two types here. To study this, the researchers used very advanced mass spectrometric techniques carried out together by Associate Professor Thomas Jørgensen and PhD student Simon Mysling at SDU. Associate Professor Jørgensen's group consists of world experts in hydrogen-deuterium exchange (HDX), which can be used to study how flexible or loosely structured the different parts of a protein are. It turns out that two different oligomers with different degrees of flexibility can be present at the same time. One type admittedly 'flaps around' more than the other, but it is nevertheless very stable and does not turn into fibrils because it is unable to 'absorb' monomer proteins and thereby grow bigger. This oligomer simply clumps together to form larger oligomers, and is the dominating (and toxic) type, corresponding to the structure shown above. The other type (which only accounts for 10