Crystal structure reveals how minor variations make receptor proteins activate or inhibit natural killer cells
Natural killer (NK) cells are white blood cells that can detect and destroy abnormal cells, including cancer cells or cells infected by viruses. A*STAR researchers have now resolved a longstanding puzzle concerning the receptor proteins that NK cells use to distinguish between normal and abnormal cells. Their work suggests ways in which NK cells could be modified to improve their response to a range of diseases.
Killer-cell immunoglobulin-like receptors (KIRs) found on the surface of NK cells bind to proteins called human leukocyte antigens (HLAs) on the surface of blood cells. This KIR–HLA interaction allows NK cells to recognize abnormal or infected blood cells. Each NK cell carries both activating KIRs and inhibiting KIRs. "These complementary KIRs provide a yin–yang balance of signals to the NK cell that determine whether it should kill the target cell or just leave it alone," says Ren.
The activating and inhibiting KIR proteins are very similar: they differ only at three or four amino acids, suggesting that they have evolved from a common gene. This similarity also raises the question of how such small variations in structure allow these receptor proteins to trigger opposite effects on the NK cells.
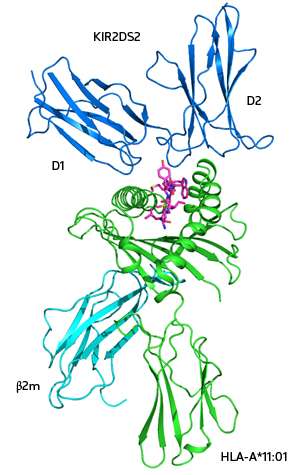
A clear way to understand the binding between the KIRs and their HLA targets is to resolve the three-dimensional structure of crystals of the complexes bound together using X-ray crystallography. To date, this has only been achieved for inhibiting KIRs. Now, Ee Chee Ren and co-workers at the A*STAR Singapore Immunology Network have managed to identify the HLA molecule that binds to a specific activating KIR, known as KIR2DS2.
Ren's team has also determined the first detailed structure of crystals of the activating KIR bound to its HLA partner, HLA-A*11:01 (see image). "The structure shows precisely how the activating KIR recognizes and binds to a different HLA than its related inhibiting KIR, and also reveals the three amino acids that differ between the two KIRs," explains Ren. However, only one of them—a tyrosine residue—seems to matter.
The researchers validated the conclusions from their crystallography studies by studying the binding of the activating KIRs to live cells.
Having clarified the different ligand-binding characteristics of activating and inhibiting receptors, the researchers now plan to investigate the detailed binding mechanism that activates the cell-killing activity of NK cells. This could advance the use of these cells in therapeutic applications, for example by enhancing their cell-killing effects against specific types of diseased cells.
More information: Liu, J., Xiao, Z., Ko, H. L., Shen, M & Ren, E. C. Activating killer cell immunoglobulin-like receptor 2DS2 binds to HLA-A*11. Proceedings of the National Academy of Sciences USA 111, 2662–2667 (2014). dx.doi.org/10.1073/pnas.1322052111