Novel drug targeting leukemia cells enters clinical trial
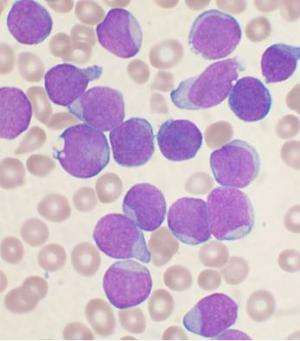
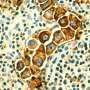
Researchers at the University of California, San Diego School of Medicine have launched a phase 1 human clinical trial to assess the safety and efficacy of a new monoclonal antibody for patients with chronic lymphocytic leukemia (CLL), the most common form of blood cancer in adults.
The new antibody targets ROR1, a protein used by embryonic cells during early development and exploited by cancer cells to promote tumor growth and metastasis, the latter responsible for 90 percent of all cancer-related deaths.
Because ROR1 is not expressed by normal adult cells, scientists believe it is a biomarker of cancer cells in general and cancer stem cells in particular. Because it appears to drive tumor growth and disease spread, they believe it also presents an excellent target for anti-cancer therapy.
Developed at UC San Diego Moores Cancer Center by Thomas Kipps, MD, PhD, who holds the Evelyn and Edwin Tasch Chair in Cancer Research, and colleagues, the antibody is called cirmtuzumab (also known as UC-961). In previous animal studies, Kipps' team reported that ROR1 is singularly expressed on CLL and also on a variety of different cancers, including cancers of the breast, pancreas, colon, lung and ovary. In mouse models of CLL, ROR1 acts as an accelerant when combined with another oncogene to produce a faster-growing, more aggressive cancer.
Cirmtuzumab was developed under the auspices of the California Institute for Regenerative Medicine's HALT leukemia grant awarded to Dennis Carson, MD, principal investigator, and Catriona Jamieson, MD, PhD, co-principal investigator to develop six distinct therapies against cancer stem cells. Kipps led one of the six projects and generated antibodies against ROR1, leading to the cirmtuzumab trial in patients with CLL.
"The primary goal of this phase I clinical trial is to evaluate whether cirmtuzumab is a safe and well-tolerated cancer stem cell-targeted agent in patients with CLL," said Jamieson, chief of the Division of Regenerative Medicine, associate professor of medicine, director of stem cell research at UC San Diego Moores Cancer Center, deputy director of the Sanford Stem Cell Clinical Center and a principal investigator of the cirmtuzumab clinical trial.
Michael Choi, MD, assistant clinical professor of medicine and co-principal investigator of the clinical trial said, "The trial will involve patients with relapsed or refractory CLL, who will receive an intravenous infusion every 14 days at Moores, followed by regular monitoring and clinic visits to assess efficacy and identify and manage any adverse effects. Initial treatment is planned for two months."