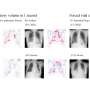
FDG-PET/CT shows promise for breast cancer patients younger than 40
Researchers at Memorial Sloan Kettering found that PET/CT imaging of patients younger than 40 who were initially diagnosed with stage I–III breast cancer resulted in change of diagnosis. As reported in the October issue of The Journal of Nuclear Medicine, while guidelines recommend FDG-PET/CT imaging only for women with stage III breast cancer, it can also help physicians more accurately diagnose young breast cancer patients initially diagnosed with earlier stages of the disease.
Assessing if and how far breast cancer has spread throughout the body is what doctor's refer to as staging. Most women nowadays are diagnosed at earlier stages, meaning stage 1 or 2 of possible 4 stages (stated Christopher Riedl, MD). Current National Comprehensive Cancer Network (NCCN) guidelines consider systemic FDG-PET/CT staging for only stage III breast cancer patients. More recently it has been debated whether factors other than stage should be considered in this decision. One such factor is patient age, as young breast cancer patients often have more aggressive tumors. In this study, a team of researchers from Memorial Sloan Kettering Cancer Center in New York evaluated for the first time the impact of FDG PET/CT staging specifically in a young patient cohort. The study suggests that breast cancer patients under the age of 40 may benefit from systemic staging with FDG PET/CT at earlier stages than NCCN guidelines suggest.
"Proper staging right after the breast cancer has first been diagnosed will help doctors make the right treatment decisions. And figuring out which breast cancer patients will benefit most from this 'advanced staging' with FDG PET/CT helps us to improve patient care while avoiding unnecessary tests," stated Christopher Riedl, MD, one of the team's lead researchers. "Our data suggest that women younger than 40 may benefit from PET/CT staging at earlier stages than doctors previously believed."
The study included 134 patients with initial diagnoses of stage I to IIIC breast cancer; those with signs of distant metastases or with prior malignancy were excluded. PET/CT findings lead to upstaging to stage III or IV in 28 patients (21%). Unsuspected extra-axillary regional nodes were found in 15/134 (11%) and distant metastases in 20/134 (15%), with 7/134 (5%) demonstrating both. PET/CT revealed stage IV disease in 1/20 (5%) patients with initial clinical stage I, 2/44 (5 %) stage IIA, 8 /47 (17 %) stage IIB, 4/13 (31%) stage IIIA, 4/8 (50%) of IIIB, and 1/2 (50%) of stage IIIC patients. All 20 patients upstaged to stage IV were histologically confirmed. Four synchronous thyroid and 1 rectal malignancies were identified.
"Future NCCN guidelines for initial staging of breast cancer patients may need to consider other factors in addition to clinical stage. This study provides further evidence that molecular imaging and nuclear medicine can help us make better cancer staging and treatment decisions," said Gary Ulaner, MD, PhD, assistant professor at Memorial Sloan Kettering. "Of course, our findings should still be confirmed in a prospective trial," he added. "Our next step will be to look at factors other than patient age to understand which breast cancer patients benefit most from FDG-PET/CT."
More information: "Retrospective analysis of FDG PET/CT for staging asymptomatic breast cancer patients below 40 years of age" Journal of Nuclear Medicine, 2014.