New data suggest little benefit of adding heart valve repair to bypass surgery in patients with coronary heart disease
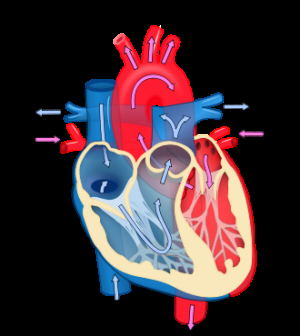
The addition of mitral valve (MV) repair (a valve of the heart) to coronary artery bypass grafting (CABG), a type of open-heart surgery, did not result in significant benefit to the patient and was associated with increased risk of neurological events. Therefore, the routine addition of MV repair to CABG in patients with moderate IMR did not demonstrate a clinically meaningful advantage.
The Cardiothoracic Surgical Trials Network (CTSN) is reporting results for the first time from a clinical trial of patients who have a complication of coronary heart disease known as moderate ischemic mitral regurgitation (IMR). Study findings were presented today at the American Heart Association's Scientific Sessions 2014 and published simultaneously in the New England Journal of Medicine.
"The results of this study suggest there is little benefit to what is often considered a routine addition to the open-heart procedure of CABG for patients with moderate IMR," said senior study author Robert Michler, M.D., professor and chairman, Department of Cardiothoracic and Vascular Surgery, Montefiore Medical Center and Albert Einstein College of Medicine, and co-director of The Montefiore Einstein Center for Heart and Vascular Care. "Although the one-year data are very compelling, long-term follow-up is necessary to determine whether the lower prevalence of IMR seen at one year will translate into any clinical benefit for patients undergoing mitral valve repair."
IMR occurs when blood backflows into the left atrium from the left ventricle of the heart due to improper closure of the MV. The condition often develops as a complication of a heart attack and subsequent enlargement of the left ventricle, the heart's main pumping chamber. Functional IMR affects 1.6 million to 2.8 million patients in the U.S. and is associated with a doubling in mortality among patients with mild or greater degrees of mitral regurgitation after a heart attack. The optimal treatment of IMR remains controversial and unclear.
"We have not had clear evidence for treatment, so recommendations vary greatly from surgeon-to-surgeon, cardiologist-to-cardiologist and institution-to-institution with perhaps one-third opting for valve repair in addition to CABG," said Peter K. Smith, M.D., professor and division chief, Cardiovascular and Thoracic Surgery, Duke University School of Medicine, and the study's primary author. "This study provides some needed clarity, but it's also important to note we must keep following these patients beyond a year, because their outcomes may change over time."
The prospective, multi-center, controlled clinical trial randomly assigned 301 patients with moderate IMR to CABG alone or CABG with MV repair. The primary endpoint was left ventricular end systolic volume (LVESVI) at one-year, assessed using a Wilcoxon rank sum test categorizing deaths as the lowest LVESVI rank.
One-year mortality was 6.7 percent in CABG/MV repair patients versus 7.3 percent in CABG patients. There were no observed differences in MACCE, death, readmissions, functional status or quality-of-life at one-year.
"This comparative effectiveness trial provides insights that should be practice-changing," said Annetine Gelijns, Ph.D., professor and chair of the Department of Population Health Science and Policy at Icahn School of Medicine at Mount Sinai, who is the corresponding author for the NEJM study and principal investigator for the Data and Clinical Coordinating Center (DCC) at Mount Sinai for the NIH-sponsored CTSN.
"There was equivalent and impressive positive remodeling of the left ventricle in both the CABG and CABG-plus-MV repair groups," said study co-author John Puskas, MD, Professor of Cardiovascular Surgery at Icahn School of Medicine at Mount Sinai and Chair of the Department of Cardiovascular Surgery at Mount Sinai Beth Israel who serves on the Steering Committee of CTSN. "The excellent 1-year survival shows that surgical performance by the sites was outstanding adding weight to our findings. Further follow-up and sub-analyses of these patients will help us understand whether the risk-benefit ratio is in favor of mitral valve repair in some of these patients or whether most should have CABG alone."
The CTSN, which is funded by NIH and CIHR, has eight core clinical centers in the U.S. and Canada, including Cleveland Clinic Foundation, Columbia University, Duke University, Emory University, Montefiore Einstein Heart Center, Montreal Heart Institute, University of Virginia, University of Pennsylvania, and 19 Consortium sites. Icahn School of Medicine at Mount Sinai, is where the CTSN's DCC is based which directs each of its clinical trial's design, methodologies, ethical research concerns, study data, analysis and reporting.
More information: This study was presented as the Late-Breaking Clinical Trial (Abstract 49403): The Surgical Treatment of Moderate Ischemic Mitral Regurgitation: A Randomized Clinical Trial From The Cardiothoracic Surgical Trials Network.