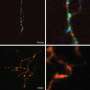
A single protein activates the machinery needed for axon growth and holds the axons together for collective extension
During brain development, neurons extend projections called axons to connect with other neurons. Axons from groups of neurons with the same function tend to extend together, but the mechanisms involved in keeping the growing axons in contact for collective extension have been unclear. Masatoshi Takeichi, Shuichi Hayashi and colleagues from the RIKEN Center for Developmental Biology and RIKEN Quantitative Biology Center have now revealed that the protein protocadherin-17 (Pcdh17) plays a crucial role in this coordinated axon growth and correct development of the nervous system.
The protocadherin family of proteins is involved in regulating cell interactions and movement, and some of these proteins have been linked to brain disorders. Pcdh17 is expressed in the amygdala—small areas of the brain that regulate emotion and social behavior.
Takeichi, Hayashi and their colleagues found that when the gene that encodes Pcdh17 was deleted in mice, fewer axons extended from the amygdala, and those that did failed to follow the usual side-by-side path. Too much Pcdh17, however, caused axons from different groups to interact anomalously. By manipulating the structure of the protein, the researchers showed that Pcdh17 molecules in neighboring axons normally bind to each other to hold extending axons together.
The team also looked for other proteins that interact with Pcdh17 inside cells. This search identified members of the five-protein complex known as WAVE. This complex controls actin polymerization—a process in which the protein skeleton of the cell is extended to allow cell migration. The WAVE complex is normally localized to structures called lamellipodia, which are found at cell edges and are required for cell movement.
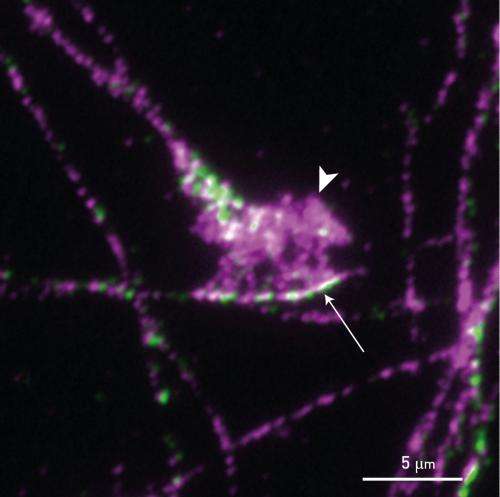
"On interacting with Pcdh17, the WAVE complex becomes localized to cell-to-cell contacts and converts these sites into motile structures," explains Takeichi. This localization was observed only at axon-to-axon contacts (Fig. 1). "We hypothesize that the motility of growth cones at the leading ends of extending axons is enhanced by the Pcdh17–WAVE complex."
The findings show that Pcdh17 plays a role in both holding groups of axons together as they grow, and in recruiting the cellular machinery required for extension.
"We discovered a mechanism by which axons extend together. A deficiency in this process may cause brain defects," says Takeichi. "Some of the other protocadherin proteins, such as Pcdh19, have been linked to disorders such as epilepsy and mental retardation in humans. We are now analyzing the cellular and molecular backgrounds of such diseases using mouse models."
More information: Hayashi, S., Inoue, Y., Kiyonari, H., Abe, T., Misaki, K., Moriguchi, H., Tanaka, Y. & Takeichi, M. "Protocadherin-17 mediates collective axon extension by recruiting actin regulator complexes to interaxonal contacts." Developmental Cell 30, 673–687 (2014). DOI: 10.1016/j.devcel.2014.07.015