Toward a cocaine vaccine to help addicts kick the habit
In their decades-long search for vaccines against drugs of abuse, scientists have hit upon a new approach to annul cocaine's addictive buzz. They report in the ACS journal Molecular Pharmaceutics that their strategy, which they tested on mice, harnesses a bacterial protein to trigger an immune system attack on the drug if it enters the body. This response could dull cocaine's psychotropic effects and potentially help users of the drug kick the habit.
Kim D. Janda and colleagues note that according to the 2011 National Survey on Drug Use and Health, an estimated 1.4 million people in the U.S. took cocaine. A number of therapies are available to help drug abusers quit. But addiction is extremely tough to beat. So some scientists are working on vaccines to neutralize the high-inducing effects of recreational drugs. Although vaccines are normally associated with fighting bacterial or viral infections, they can also be designed to recruit the body's immune system to recognize non-microbial substances such as drugs. None so far are widely effective, so Janda's team set out to try a new approach.
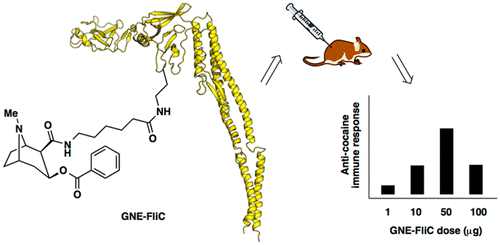
The researchers took a safe bacterial protein called flagellin that has already been incorporated in other vaccines and modified it to boost the immune response to cocaine. They tested the compound in mice and found that it worked better than a vaccine candidate they'd developed previously. The strategy, the researchers conclude, opens up a new avenue for designing vaccines against drugs of abuse.
More information: "Flagellin as Carrier and Adjuvant in Cocaine Vaccine Development" Mol. Pharmaceutics, Article ASAP. DOI: 10.1021/mp500520r
Abstract
Cocaine abuse is problematic, directly and indirectly impacting the lives of millions, and yet existing therapies are inadequate and usually ineffective. A cocaine vaccine would be a promising alternative therapeutic option, but efficacy is hampered by variable production of anticocaine antibodies. Thus, new tactics and strategies for boosting cocaine vaccine immunogenicity must be explored. Flagellin is a bacterial protein that stimulates the innate immune response via binding to extracellular Toll-like receptor 5 (TLR5) and also via interaction with intracellular NOD-like receptor C4 (NLRC4), leading to production of pro-inflammatory cytokines. Reasoning that flagellin could serve as both carrier and adjuvant, we modified recombinant flagellin protein to display a cocaine hapten termed GNE. The resulting conjugates exhibited dose-dependent stimulation of anti-GNE antibody production. Moreover, when adjuvanted with alum, but not with liposomal MPLA, GNE-FliC was found to be better than our benchmark GNE-KLH. This work represents a new avenue for exploration in the use of hapten-flagellin conjugates to elicit antihapten immune responses.