January 28, 2015 report
Everything you wanted to know about mitochondrial mutations but were afraid to ask
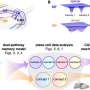
(Medical Xpress)—In a recent post we traced mechanisms of mitochondrial quality control in the nervous system back to their origins in the womb, and hinted that there would soon be more to come on this topic. Conveniently, a special issue of the journal Molecular Health Research came along and did some of the dirty work for us. In particular, several of the papers address an important question regarding mitochondrial mutations and heteroplasmy that many involved in the brave new business of three-parent embryos are likely afraid to ask. Now that we understand the importance of mitochondria to what we might broadly call "fertilization outcome", the question is do we really know enough about mitochondria to hand craft artisanal children from aged or otherwise reproductively questionable feedstock in the way many imagine?
For those families fortunate enough to be part of Apple's new eggs on ice program, some of the incremental risk incurred in passing on suboptimal mitochondrial can be deferred. For the rest of us, especially the moms already carrying mitochondrial disease genes, there is an important concept known as mutational load that we need to get more familiar with. Preimplantation genetic diagnosis, where a cell is removed for diagnosis from the early 5-10 cell embryo, is one way to assess mtDNA mutant load. This is a bit more invasive than just checking on a turkey in the oven because even if the embryo, minus its pound of flesh, is returned to the womb its unwritten 'do not tamper' sign has been irrevocably disturbed.
What researchers have found, is that not only does mutation load vary significantly among individual blastomeres in early embryo, but it is also skewed in the individual organs that are eventually established. The granddaddy of mutation load investigations was done by Sharpley et. al., who followed some 500 mice over the course of 14 generations. This particular study gave us some further deflating news for trying to make three-parent embryos: heteroplasmy itself, that is to say just having different kinds of mitochondria floating around, can cause all kinds of abnormalities. These researchers found that as long as a mouse's mitochondria were largely homoplasmic, it didn't really matter if they had so-called NZB or a 129 mutations in their mtDNA. However, when heteroplasmy was bumped up closer to 50% NZB and 50% 129, there were huge behavioral and neural effects.
One important observation in these 50/50 mice was that the 129 mtDNA is lost in organs like the liver and kidney. In these organs an increase in the demand for energy and therefore ATP synthesis is met through biogeneis as opposed to places like the brain, heart and skeletal muscle where it is met primarily through phosphorylation of respiratory complexes. Of note, regulation in the brain and muscle occurs through a much higher dynamic range, and curiously, the NZB and 129 haplotypes were not segregated here like they were in liver and kidney. In eggs and ovarian tissues it was the NZB mtDNA that was preferentially lost over the course of several generations to restore homoplasmy.
The intuition that different parts of the embryo distinguish between mtDNA haplotypes raises the question of whether it is the embryo that is controlling the spatial activities of the mitochondria or if it is the mitochondrial that control and geometrically define the egg. Either way, it is a self-evident truth that the job of building the nervous system—the organ where the effects of mtDNA issues are most evident—falls squarely on the egg. In order to actively create composite offspring that are free of the diseases which energetically cripple the brain, as opposed to just passive selection/abortion for misprints, there are currently two main approaches that are on the socio-political table. Neither method is non-invasive, and neither is guaranteed to eliminate all the bad guys from the embryo's mitochondrial pool.
The first method is called metaphase II spindle transfer and it is done before fertilization. Here the entire nuclear-spindle structure from the egg of the mutantDNA carrier is removed and put into a recipient donor egg (presumably containing all optimal mitochondria) whereupon it and the sperm are introduced to each other. Although it would seem that the cytoplasmic order and thin spindle fibers would get disrupted in this handling as much as one would expect an intricate spider web to be diminished if we tried to move it to another tree, the procedure does seem to be able to get things to the next level. The other method is called pronuclear transfer, and here the pronuclei from a fertilized egg is transferred into a recipient donor zygote.
Beyond the simple practicalities of who all to put on a new age birth certificate lies the tougher question of how to go about matching donors and recipients in light of the issues hinted at above. Like HLA matching, we clearly don't know all the variables at play, and have far less understanding of the general principles involved. In this void, perhaps it is best to take a step out and look at some new discovers in the larger fold of the mutations, the kinds of RNAs encoded by mitochondria, and even the ribosome itself.
The Journal of Theoretical Biology recently published a sweeping hypothesis regarding ribosomes and origin of life. Although it has already garnered much attention, so far it has furnished little in the way of a larger community consensus. The authors sought to answer the question of what it is that DNA is really doing in much the same way that we must ask that question here about mitochondria. The author's appear to have found sequence-based evidence that ribosomal RNA "represents a primitive genome that encodes the instructions needed to direct ribosomal replication, translation, and self organization." This grand conception, in addition to other recent discoveries demonstrating extra-ribosomal protein synthesis, draws attention to some of the shortcomings of the foundational RNA world hypothesis, and forces us to re-orient our perspective regarding the primacy of RNA vs proteins.
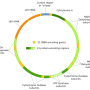
For us, the paper's key observation that tRNAs are encoded in an overlapping manner within rRNA in a fashion similar to how they are encoded in the mitochondria of all metazoans, may be of particular interest. We can also point to another happenstance similarity in the functional organization of tRNA and rRNA sequences, again not necessarily historical for the evolution of mitochondria. That is that both strands of mtDNA are transcribed into large polycistronic rRNA and mRNA transcripts punctuated by tRNAs. The tRNAs are processed out (probably not like introns are, but again, perhaps reminiscent) before the mRNAs are translated on mitochondrial 55S ribosomes.
While these specific kinds of editing processes hold long-standing interest, RNA editing in general has only recently come to be seen as common. We now know that not ony does it occur in mitochondria, but in the squid nervous system the majority (some 57,000 spots) of transcripts are recoded by RNA editing. The comparatively small nucleoids of mitochondria are steeped in the art of RNA. Considering that the proteins of the respiratory chain in all cells are the product of 2 genomes—nuclear & mitochondrial—the larger spectrum of optimal matching and RNA control are important for issues beyond just reproductive compatibility.
The need for on-the-fly nuclear genome matching would seem to ensure that we needn't ever worry that the three-parent-embryo issue would result in some kind of government-issue or standardization of optimal mitochondria. On the other hand, indefinite mitochondrial mutation also means it is naive to think we could ever fully eliminate the so-called bad mitochondria from any germline because new polymorphism is continually generated as a natural byproduct of the system. Regardless of what different governments decide to do with this issue in the short term, the longer term will undoubtedly be informed primarily by hard science and living anecdote.
More information: The mitochondrion, its genome and their contribution to well-being and disease, Mol. Hum. Reprod. (2015) 21 (1): 1-2. molehr.oxfordjournals.org/content/21/1/1.full
© 2015 Medical Xpress