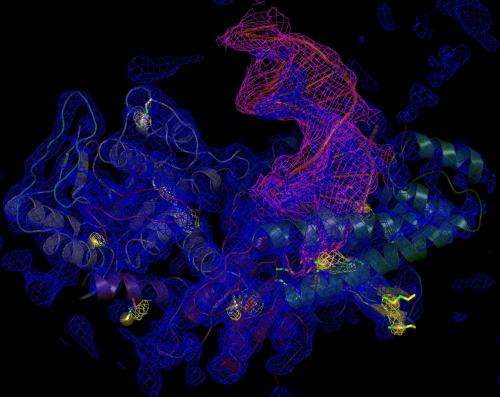
3-D snapshot of protein highlights potential drug target for breast cancer
The genome of a cell is under constant attack, suffering DNA damage that requires an army of repair mechanisms to keep the cell healthy and alive. Understanding the behavior of the enzymes defending these assaults helps determine how - and where - cancer gets its foothold and flourishes. New research published in an Advance Online Publication of Nature Structural & Molecular Biology shows that one of these enzymes - human DNA polymerase theta (POLQ) - may be a promising drug therapy target for inhibiting breast cancer.
"The human genome encodes more than 15 different polymerases, which are the essential enzymes that copy our genetic material," says Karl Zahn, Ph.D., University of Vermont postdoctoral associate and first author on the study. "POLQ is known to serve a specialized role - it can perform some tasks that other polymerases are unable to do, including the repair of certain toxic double-strand breaks in the DNA, which can induce genomic instability if left unrepaired."
This genomic instability can sometimes lead to cancer, especially the types of cancer that are most difficult to treat.
Using X-ray crystallography, Zahn and University of Vermont structural biologist Sylvie Doublié, Ph.D., and colleagues determined the first crystal structures of POLQ - recently highlighted as the only DNA polymerase overexpressed in certain breast and ovarian cancers - providing a visual of the DNA repair process.
"The field focused on this enzyme is evolving," says Doublié, senior author on the paper, which is one of several POLQ-related studies published in the last month. The team's work provides a first step in determining how to feasibly pursue POLQ as a cancer drug target.
"Having this new structure really helps inform this process," says Doublié.
While already found in breast, ovarian, lung and oral cancers, POLQ is believed to play a role in a number of additional cancers, so the impact of the group's finding could be far-reaching in terms of new therapies.
Doublié and Zahn's coauthor Richard Wood, Ph.D., of the Department of Epigenetics & Molecular Carcinogenesis at the University of Texas MD Anderson Cancer Center, and colleagues are in the process of examining candidate drug-like molecules that might act specifically on POLQ without inhibiting other necessary polymerases to limit side effects in cancer patients.
In addition to Doublié, a professor of microbiology and molecular genetics, Zahn, and Wood, coauthors on the study include April Averill, University of Vermont senior research technician in microbiology and molecular genetics, and Pierre Aller, Ph.D., of Diamond Light Source, Didcot, Oxfordshire, UK.
More information: Human DNA polymerase θ grasps the primer terminus to mediate DNA repair, Nature Structural & Molecular Biology (2015) DOI: 10.1038/nsmb.2993