Study shows feasibility of using gene therapy to treat rare immunodeficiency syndrome
In a small study that included seven children and teens with Wiskott-Aldrich syndrome, a rare immunodeficiency disorder, use of gene therapy resulted in clinical improvement in infectious complications, severe eczema, and symptoms of autoimmunity, according to a study in the April 21 issue of JAMA, a theme issue on child health.
Wiskott-Aldrich syndrome (WAS) is caused by loss-of-function mutations in the WAS gene. The condition is characterized by thrombocytopenia (low platelet count), eczema, and recurring infections. In the absence of definitive treatment, patients with classic WAS generally do not survive beyond their second or third decade of life. Partially human leucocyte antigen (HLA) antigen-matched allogeneic (donated from another individual) hematopoietic stem cell (HSC) transplantation is often curative, but is associated with a high incidence of complications. Gene therapy based on transplantation of autologous (from the same individual) genecorrected HSCs may be an effective and potentially safer alternative. This procedure involves the removal and treatment of the patient's own blood stem cells, and their return to the patient by intravenous injection.
Marina Cavazzana, M.D., Ph.D., of Necker Children's Hospital, Paris, France, and colleagues assessed the outcomes and safety of autologous HSC gene therapy in patients with Wiskott-Aldrich syndrome. Gene-corrected autologous HSCs were infused in 7 patients (age range, 0.8-15.5 years) with severe Wiskott-Aldrich syndrome lacking HLA antigen-matched related or unrelated HSC donors. Patients were enrolled in France and England and treated between December 2010 and January 2014. Follow-up of patients in this intermediate analysis ranged from 9 to 42 months.
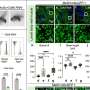
Among 6 of the 7 patients, there was clinical improvement after gene therapy, which was well tolerated. One patient died of preexisting, treatment-refractory infectious disease. In the 6 surviving patients, the infectious complications resolved after gene therapy, and prophylactic (preventative) antibiotic therapy was successfully discontinued in 3 cases. Severe eczema resolved in all affected patients, as did signs and symptoms of autoimmunity.
No severe bleeding episodes were recorded after treatment, and at last follow-up, all 6 surviving patients were free of blood product support. Hospitalization days were reduced from a median of 25 days during the 2 years before treatment to a median of 0 days during the 2 years after treatment.
The authors note that the interpretation of the results of this type of study is constrained by the small number of patients. "We therefore cannot draw conclusions on long-term outcomes and safety. Further follow-up of these patients and those reported in a similar study last year, together with additional clinical trials of this therapy, are therefore necessary."
In an accompanying editorial, Harry L. Malech, M.D., of the National Institutes of Health, Bethesda, Md., and Hans D. Ochs, M.D., of the University of Washington and Seattle Children's Research Institute, Seattle, write that this study provides strong evidence that this type of gene therapy achieves substantial restoration of immune function associated with prolonged clinical benefit to patients with severe WiskottAldrich syndrome.
They add that the impressive clinical response seen in the study was achieved in the context of a long line of research by many groups of investigators striving toward the goal of clinically beneficial gene therapy. "Taken together, the available evidence demonstrates substantial sustained clinical benefit following gene therapy for certain diseases."
"At a time when many are championing personalized medicine, no advance is as representative of that fundamental biological approach as gene therapy."
More information: Outcome following Gene Therapy in Patients with Severe Wiskott-Aldrich Syndrome, JAMA, 2015. doi:10.1001/jama.2015.3253
Editorial: doi:10.1001/jama.2015.2055