Team reports promising results of phase 1/2 trial of rociletinib in EGFR lung cancer
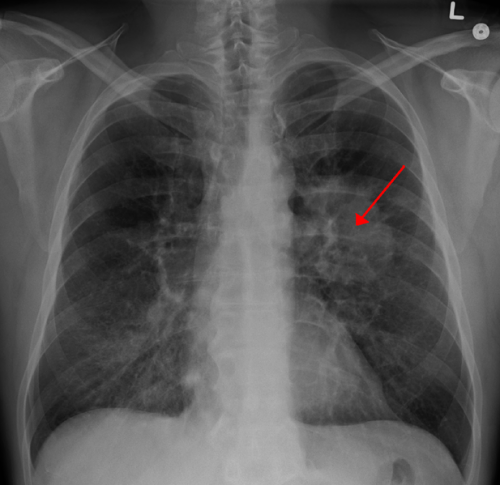
The New England Journal of Medicine reports results of a multi-center phase I/II study of the investigational anti-cancer agent rociletinib (CO-1686) in patients with EGFR-mutant non-small cell lung cancer (NSCLC) that had progressed after previous treatment with EGFR inhibitors. Responses were seen in 59 percent of evaluable patients with the T790M mutation (n=46). In this same population, median progression-free survival (PFS) at the time of analysis was 13.1 months; these data continue to mature.
Mutations in the epidermal growth factor receptor (EGFR) gene occur in about 10-15 percent of lung cancers in the United States and Europe and in 30-35 percent of lung cancers in Asia. The mutation locks the EGFR into an active state driving the growth of the cancer, while also making tumors exquisitely sensitive to existing drugs such as erlotinib, gefitinib or afatinib, which function as EGFR inhibitors. Unfortunately, after a median of 9-13 months, lung cancers begin to evolve resistance to these drugs. Approximately 50-60 percent of disease progression in this setting occurs through a further, specific mutation of the EGFR gene, named T790M. Results show that it is specifically in tumors with this secondary mutation, or "escape mechanism", of T790M that rociletinib is most active. There are no currently approved therapies for T790M+ NSCLC.
"This is a potential game changer for lung cancer patients whose disease is driven by EGFR mutations. Previously, once tumors learned to evade treatment with first-line EGFR inhibitors, we had no second targeted treatment. With these promising results, it is looking extremely likely that we now have a therapy that will keep people alive, well and in the game," says D. Ross Camidge, MD, PhD, director of thoracic oncology at the University of Colorado Cancer Center and the study's senior author. The drug rociletinib is owned by Clovis Oncology, Inc., which is based in Boulder, Colorado. Due to strong early results, the drug received the new FDA 'Breakthrough Status' designation, developed in part by Colorado's Senator Michael Bennet as a means to accelerate the approval process for very promising new medical treatments.
In the current study, patients were initially given free base and later hydrogen bromide salt formulations of the drug rociletinib in ongoing 21-day cycles. The hydrogen bromide salt produced better absorption and higher drug exposure in patients and was adopted as the sole form moving forward.
The most common side effect was hyperglycemia occurring in 47 percent of patients treated with a therapeutic dose, of whom 22 percent experienced Grade 3; no hyperglycemia events led to drug discontinuation. This side effect is caused by a specific metabolite of rociletinib, which has activity against the insulin-like growth factor 1 receptor and insulin receptor. The expansion phase of the trial is ongoing and continues to evaluate the efficacy of the drug in a larger population of patients.
"The rociletinib study is an elegant example of how far we have come in the last 10 years in understanding the biology of NSCLC and applying it directly in the clinic. To be able to test patients' tumors to determine the best drug for their specific situation - not only at diagnosis, but now at acquired drug resistance too - this represents precision medicine in real life. It brings us one step closer to changing lung cancer from a deadly diagnosis into a chronic and treatable condition," says Lecia V. Sequist, MD, MPH, of the Massachusetts General Hospital (MGH) Cancer Center, principal investigator of the trial and corresponding author of the NEJM paper.
In addition to the CU Cancer Center and the UCLA David Geffen School of Medicine, participants in the clinical trial include Massachusetts General Hospital, Harvard Medical School, Dana-Farber Cancer Institute, Gustave Roussy Cancer Campus, Stanford Cancer Institute, Karmanos Cancer Institute, Institute Gustave Roussy, MD Anderson Cancer Center, Peter MacCallum Cancer Centre, Medical University of Gdansk, and Clovis Oncology.
"On average, we're talking about more than a year of disease control in patients who were without targeted treatment options," says Jonathan Goldman, MD of the UCLA David Geffen School of Medicine, one of the other investigators on the study. "In patients with such advanced disease, who had sometimes been through multiple priors rounds of therapies, to see disease control like this is very gratifying."