Researchers unveil new gene subgroup in prostate cancer
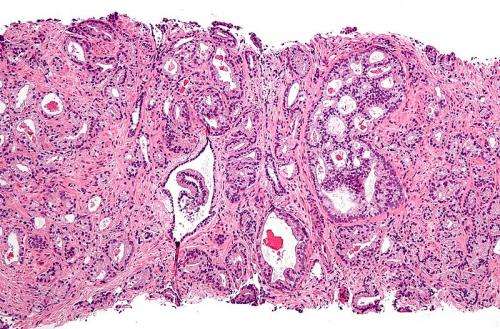
Prostate cancer researchers have drawn a molecular portrait that provides the first complete picture of localized, multi-focal disease within the prostate and also unveils a new gene subgroup driving it.
The discoveries, published online today in Nature Genetics, are a further step along the road to personalizing prostate cancer medicine say study co-leads, Dr. Robert Bristow, a clinician-scientist at Princess Margaret Cancer Centre, and Dr. Paul Boutros, an investigator at the Ontario Institute for Cancer Research.
"Our research shows how prostate cancers can vary from one man to another - despite the same pathology under the microscope - as well as how it can vary within one man who may have multiple tumour types in his prostate," says Dr. Bristow. He goes on to say, "these sub-types may be important to determining the response to surgery or radiotherapy between patients."
The study involved molecular profiling of 74 patients with Gleason Score 7 index tumours. (Gleason is the classification system used to evaluate aggressiveness in prostate tumours). Of these, whole-genome sequencing was done on 23 multiple tumor specimens from five patients whose prostates were removed at surgery. By carefully analyzing the genetics of each focus of cancer within each prostate, the researchers could assign "aggression scores" to each cancer which revealed that even small cancers can contain aggressive cells capable of altering a patient's prognosis.
Dr. Boutros, explained that the more detailed analysis clearly identified that two members of the MYC cancer gene family were at play in disease development, and that one of them - "C-MYC" - was the culprit driving aggressive disease. The other one - "L-MYC" - is already known to be implicated in lung and other cancers.
"This discovery of a new prostate cancer-causing gene gives researchers a new avenue to explore the biology of the disease and improve treatment," says Dr. Paul Boutros, a principal investigator at the Ontario Institute for Cancer Research.
"By showing that mutations in prostate cancer vary spatially in different regions of a tumour, this study will aid in the development of new diagnostic tests that will improve treatment by allowing it to be further personalized."
Dr. Bristow says about half of all prostate cancer patients have either C-MYC or L-MYC mutations, but never both: "Our findings suggest we are getting closer to subtyping prostate cancer based on which gene is present to determine a patients' disease aggression in terms of the risk of spread outside the prostate gland at time of treatment. In developing this research tool into a clinical test within three years, we hope to inform doctors and patients about specialized treatments for each prostate cancer patient."
Drs. Bristow and Boutros co-lead the Canadian Prostate Cancer Genome Network (CPC-GENE) Sequencing Project. Dr. Bristow is a Professor in the Departments of Radiation Oncology and Medical Biophysics, University of Toronto, where Dr. Boutros is an Assistant Professor, in Department of Medical Biophysics, University of Toronto.
In November, 2014, the long-time collaborators published earlier findings in Lancet Oncology about the genetic test they have developed to identify which men are at highest risk for their prostate cancer to come back after localized treatment with surgery or radiotherapy.
More information: Spatial genomic heterogeneity within localized, multifocal prostate cancer, DOI: 10.1038/ng.3315