June 11, 2015 report
Study reveals gene mutation that confers resistance against prion disease
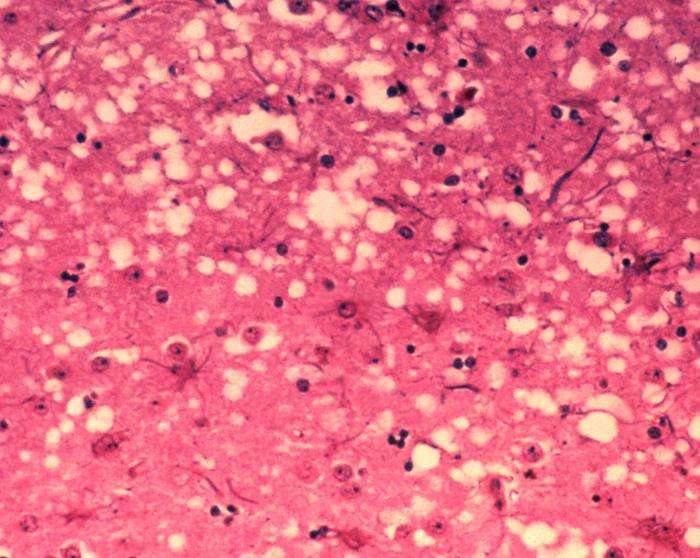
(Medical Xpress)—A team of researchers with the UCL Institute of Neurology in the U.K. with assistance from a pair of researchers in New Guinea, has found a gene mutation that confers complete resistance against a type of prion disease. In their paper published in the journal Nature, the team describes how they studied the genes of people believed to be resistant to Kuru, a type of prion disease and how that led to the discovery of the resistant gene mutations. Glen Telling with Colorado State University offers a News & Views piece on the work done by the team in the same journal edition.
Prion diseases such as "mad cow" are confounding ailments—there is no virus or bacteria that can be identified, instead, those afflicted suffer from misfolded proteins that lead to brain or other neurological deterioration—to date, there is no cure for them. Because they are still relatively new to science, information is still being obtained in how they come about and ultimately how to treat such patients. Thanks to the efforts of the researchers with this new effort, there may be a way to prevent people from getting such diseases in the first place.
To learn more about such diseases, the researchers enlisted the assistance of some members of the Fore people of Papua, New Guinea—half a century ago they were experiencing an outbreak of the prion disease Kuru—the disease was propagated, as it turned out, by cannibalism. Some of those people appeared to be immune to the disease and helped to repopulate devastated communities. It was their descendants that assisted with this new research.
After careful genetic analysis, the researchers discovered two genetic mutations (variants) in the volunteers' DNA that appeared to be connected to immunity from Kuru. Mice genetically modified to be susceptible to the disease gained resistance when one or the other of the variants was caused to come about in their genes—when both variants were introduced, the mice became one hundred percent resistant.
Moving forward, the researchers plan to see if they can find out how or why the genetic mutation causes resistance and of course, seek to discern if the same technique could be used to confer resistance in humans.
More information: A naturally occurring variant of the human prion protein completely prevents prion disease, Nature (2015) DOI: 10.1038/nature14510
Abstract
Mammalian prions, transmissible agents causing lethal neurodegenerative diseases, are composed of assemblies of misfolded cellular prion protein (PrP)1. A novel PrP variant, G127V, was under positive evolutionary selection during the epidemic of kuru—an acquired prion disease epidemic of the Fore population in Papua New Guinea—and appeared to provide strong protection against disease in the heterozygous state2. Here we have investigated the protective role of this variant and its interaction with the common, worldwide M129V PrP polymorphism. V127 was seen exclusively on a M129 PRNP allele. We demonstrate that transgenic mice expressing both variant and wild-type human PrP are completely resistant to both kuru and classical Creutzfeldt–Jakob disease (CJD) prions (which are closely similar) but can be infected with variant CJD prions, a human prion strain resulting from exposure to bovine spongiform encephalopathy prions to which the Fore were not exposed. Notably, mice expressing only PrP V127 were completely resistant to all prion strains, demonstrating a different molecular mechanism to M129V, which provides its relative protection against classical CJD and kuru in the heterozygous state. Indeed, this single amino acid substitution (GV) at a residue invariant in vertebrate evolution is as protective as deletion of the protein. Further study in transgenic mice expressing different ratios of variant and wild-type PrP indicates that not only is PrP V127 completely refractory to prion conversion but acts as a potent dose-dependent inhibitor of wild-type prion propagation.
© 2015 Medical Xpress