Discovery could improve in vitro fertilization success rates for women around the world
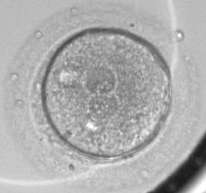
Scientists at Oregon Health & Science University, Stanford University, University of Valencia and IGENOMIX have discovered that chromosomal abnormalities in human embryos created for in vitro fertilization, or IVF, can be predicted within the first 30 hours of development at the cell-1 stage which results from the union of a female egg and male sperm.
This discovery, published online today in the journal Nature Communications, could improve IVF success rates, which has hovered around 30 to 35 percent for numerous years worldwide. It is estimated that between 50 to 80 percent of embryos created for IVF have a chromosomal abnormality and typically do not develop into a pregnancy, instead resulting in a miscarriage.
Key findings of this research, which was conducted by Shawn L. Chavez, Ph.D. and her colleagues at Stanford University and analyzed at OHSU, showed that by looking at the duration of the first mitotic phase - a short period in the cell cycle - one can identify chromosomally normal versus abnormal embryos up to approximately the 8-cell stage. Most importantly, by looking at a single cell level, researchers were able to correlate the chromosomal make-up of an embryo to a subset of 12 genes that are activated prior to the first cell division. These genes likely came from the gametes - the eggs or sperm - and can be used to predict whether an embryo is chromosomally normal or abnormal at the earliest stage of human development.
As a result of these findings, clinicians and embryologists can more quickly identify the healthiest embryo for implantation and reduce the amount of time an embryo is cultured in the laboratory prior to transfer. Embryos typically need to be implanted within three to five days of creation, which has created a challenge for the IVF field because chromosomal abnormalities may not be identified until day five or six.
"Many couples are choosing to have children later in life, and this trend is only going to continue," said Chavez, a co-author and assistant scientist in the Division of Reproductive and Developmental Sciences at the Oregon National Primate Research Center at OHSU, and assistant professor of Obstetrics and Gynecology, and Physiology and Pharmacology, in the OHSU School of Medicine. "A failed IVF attempt takes an emotional toll on a woman who is anticipating a pregnancy as well as a financial toll on families, with a single IVF treatment costing thousands and thousands of dollars per cycle. Our findings also bring hope to couples who are struggling to start a family and wish to avoid the selection and transfer of embryos with unknown or poor potential for implantation," explained Chavez.
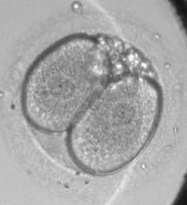
This study builds upon previous research published by Stanford University School of Medicine (Nature Communications, Dec. 2012) in which Chavez was the lead author. In the previous study, Chavez and colleagues determined that the timing intervals of the first three mitotic cellular divisions, plus the presence or absence of cellular fragmentation - a phenomenon of unknown origins that is thought to be acellular debris and affects approximately 50 percent or more of IVF embryos - can be used to largely distinguish chromosomally normal and abnormal embryos by the 4-cell stage through time-lapse imaging analysis.
The current study took advantage of significant advances in both single-cell genetic profiling and noninvasive imaging, and is the first study to combine analysis of complete chromosomal constitution, high throughput single-cell gene expression and time-lapse imaging simultaneously in the same human embryo.
This research was conducted using 117 human zygotes originating from 19 couples, with an average maternal age of 33.7 ± 4.3 years. Researchers obtained this large set of human embryos from previous IVF cycles after written informed consent from the Stanford University RENEW Biobank. Through single cell gene expression profiling - the measurement of the activity of hundreds of genes at once - Chavez and colleagues identified a subset of developmentally relevant genes that are activated prior to the first cell division using computer-assisted mathematical modeling that can be used to predict whether the embryo is chromosomally normal or abnormal.
"With assisted reproduction at an all-time high, we want to help more families achieve successful pregnancies," said Chavez. "IVF has helped countless women all over the world, and we now have the technology and research to improve a couple's chances of having a biological child of their own. This discovery can potentially increase those chances."
It is still unclear what impact longer culture periods have on embryos and genetic abnormalities and requires further investigation. Researchers say future studies should also focus on the zygote as a potential source of non-invasive biomarkers that can prospectively predict chromosomal status and avoid potential detriment(s) of prolonged embryo culture.
More information: Prediction model for aneuploidy in early human embryo development revealed by single-cell analysis, Nature Communications 6, Article number: 7601, DOI: 10.1038/ncomms8601














