July 10, 2015 report
Mitochondria control oncogenesis through metabolic reprogramming
(Medical Xpress)—Perhaps the boldest attempt to date at a single unified explanation of cancer is that offered by Albert Szent-Gyorgi in his classic book, The Living State. That was over 40 years ago. His insights on the processing of radicals in particular, and on the primal significance of electron donors and acceptors in general, still stand as remarkably compact descriptions of the constant essential features of cell function as its specific type varies.
Part and parcel to his unique view of life are the transformative events precipitated by the acquisition of atmospheric oxygen, and the cellular machinery to process it—namely, eukaryogenesis, and the mechanisms of repression and subjugation to the whole that were required in the subsequent transitions to multicellularity. Since his time, advances in genetics have afforded us a vast treasury of knowledge regarding specific mutational insults and the specific cancers they eventually lead to. Similarly, those same genetic advances have allowed us to piece together the mutualistic mergers between the bacterial precursors of eukaryotic cells and the mitochondria endosymbionts they acquired.
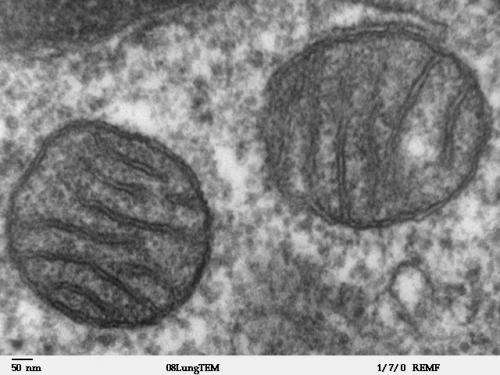
As the essential role of mitochondria in kicking off early events in the developing embryo and the ensuing differentiation of its cells is beginning to be laid bare, it is only now that the centrality of mitochondria in realizing or averting full blown tumorigenesis is coming to the fore. A recent review by Thomas Seyfried concludes that cancer is more a mitochondrial metabolic disease in accordance with the original theory of Otto Warburg, than it is the product of point or accumulated somatic mutations. The 'Warburg effect' is the observation that cancer cells produce energy by a high rate of glycolysis followed by lactic acid fermentation in the cytosol. In normal cells, there is generally a low rate of glycolysis followed by oxidation of pyruvate in mitochondria.
While there is nothing wrong with the current thinking that a rapidly expanding list of mutated 'driver' genes and resultant brand of tumor they generate constitutes an explanation, something more is needed—something to unite and simplify. The idea that somatic mutations responsible for cancer spontaneously arise through random insult or error during DNA replication in normal noncancerous stem cells is certainly not implausible. However, Warburg's conception of respiratory insufficiency (failure of the oxidative phosphorylation or OXPHOS function of mitochondria) as the origin of cancer, its phenotypic descriptions, and even somatic mutation itself, not only has simplistic appeal, it can also be tested.
Some of the primary evidence for the theory now comes from nuclear cytoplasm transfer experiments in which so-called 'cybrid' composite cells are created. Tumorigenicity was found to be suppressed when the cytoplasm of enucleated normal cells was fused with nucleated tumor cells. In other words normal cytoplasm, containing mitochondria from non-tumorigenic cells, could suppress the malignant phenotype of tumor cell. On the other hand, cybrid experiments also showed that normal cell nuclei could not suppress tumorigenesis when placed in tumor cell cytoplasm. Therefore normal nuclear gene expression, including presumptive normal tumor suppressor genes, doesn't rescue malignancy. This may not necessarily prove that every road to cancer has to pass through mitochondria any more than finding centrioles in dividing cells proves that every cell needs functioning centrioles to divide, but it does limit scope of the somatic mutation conception.
A cytoplasmic origin of cancer, as opposed to a nuclear origin, does not mean that the problem has to be the mtDNA itself. It can potentially be the result of a problem in the mitochondrial network that regulates everything from calcium levels up to the scale of cell division itself. There is however, some exciting new work that indicates disruption of essential OXPHOS proteins encoded by mtDNA induce metabolic reprogramming and the Warburg effect. In particular, it was shown that genetic silencing of cytochrome c oxidase by shRNA expression resulted in a metabolic shift to glycolysis, loss of anchorage-dependent growth, and acquisition of invasive phenotypes. This suggests that defects in the mitochondrial electron transport chain can initiate a retrograde signaling cascade that leads to tumor progression.
There is also new work at the yeast mitochondrial network level that suggests major revisions of our accepted norms at a fundamental level of cell biology may be needed. Generally, in plants and budding yeast mitochondria are mostly positioned by actin filaments, while in higher eukaryotic cells and fission yeast they are distributed by microtubules. Neurons, we might mention, do a little bit of both.To be properly inherited, fission yeast mitochondria must be reconfigured into a tubular network structure by microtubules. The intriguing thing here is that it now seems that yeast mitochondria can move and be moved without the action of any motor proteins at all. This is something we have suggested should be possible several times before, only now there is some evidence to show it. This idea stands in sharp contrast to the standard view which presumes that despite their comparatively huge and draggy footprint in the cell, mitochondria are somehow nimbly towed about by one or two miniscule motor protein tethers through a dense tangle of cytoskeleton.
It should not be that much of a stretch to go from the idea that the structure and behavior of mitochondria networks is an important part of cell division, to the idea they can control cell division, and therefore potentially control cancer in the differentiated tissues of multicellular organisms. In talking to noted medical geneticist and single gene champion Shane McKee about the new review from Seyfried, it is probably safe to say folks will be holding on to the somatic mutation theory for some time. Shane accepts that mitochondria play a significant role in cancer, but rejects the idea that they can be considered as 'the root' of cancer in the sense that the problem originates in mitochondria before it is evident in the genes.
The task ahead may be to show that all the other intangibles than mitochondria do in addition to or in support of their coming up with the standard OXPHOS deliverables—their constant fizz, fuse, and general fuss in the cytoplasm—actually directly participates in the maintainance of the noncancerous state of cellular and organismal health. While it is accepted that this activity serves to weed out defective mtDNA and potentially undesireable mtDNA heteroplasmy it also likely does a lot more. One such function would be to homogenize 'protein heteroplasmy' among mitochondria.
With perhaps a thousand or so different nuclear encoded proteins bearing mitochondrial localization sequences dispatched by cell central, it probably goes without saying that it is a major challenge to get the right protein blend in all of the cells mitochondria. This situation is no doubt amplified in the spatially extended structure of complex neurons. Fortunately, most neurons needn't worry about the extra overhead imposed by cell division, or for that matter cancer.
More information: Cancer as a mitochondrial metabolic disease, Front. Cell Dev. Biol., 07 July 2015 | dx.doi.org/10.3389/fcell.2015.00043
© 2015 Medical Xpress