July 17, 2015 report
Researchers discover web-like structures that may contribute to immune response to atherosclerosis
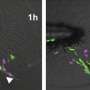
(Medical Xpress)—A small team of researchers with the Francis Crick Institute in London has identified web-like structures called neutrophil extracellular traps (NETs) that develop in the body of mice that may contribute to an inflammatory response to atherosclerosis. In their paper published in the journal Science, the group describes several experiments they conducted with mice that helped to reveal the nature of the NETs and the role they play in chronic inflammation. Matthias Nahrendorf and Filip Swirski of Harvard Medical School offer a Perspective piece on the work done by the team in the same journal article.
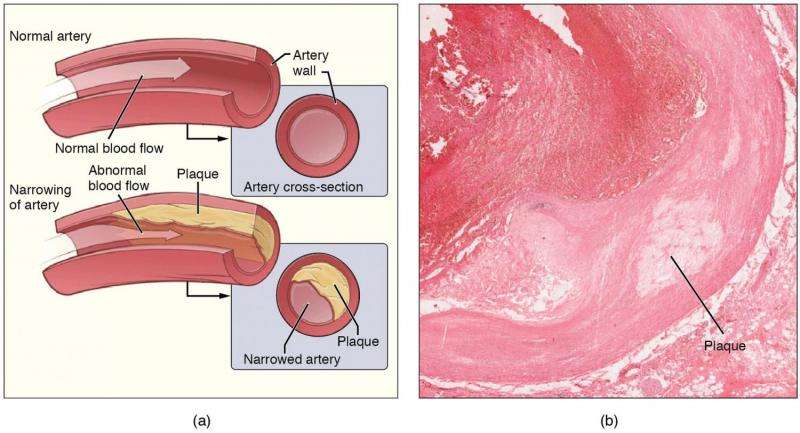
Atherosclerosis, as most people know by now is a condition that occurs when fat and other material adheres and builds up in artery walls—that's bad, of course because it can lead to heart attack or stroke. Less well known is that it can also lead to chronic inflammation, which can damage tissue in the body—this happens, prior research has shown due to an immune response. That immune response involves neutrophils and macrophages—neutrophils lead the charge and are followed by macrophages. The job of both is to counterattack things that seem dangerous and one of the results is inflammation—nature's version of a cast. Unfortunately, sometimes the apparent danger doesn't go away, such as when fat persists on arterial walls, and that can lead to chronic inflammation, which is bad, because it can cause tissue damage and other long term or permanent changes to body parts. What has not been clear is the process by which neutrophils and macrophages go about handling long term challenges. In this new effort, the researchers have found that neutroophils actually prime macrophages in response to persistent atherosclerosis.
To do their work, neutrophils expel material that hopefully kills invading microbes, but, the researchers found when the battle continues for a certain amount of time, the body sends in reinforcements in the form of histones, DNA and myeloperoxidas to help the neutrophil elastase—together they make up the NETs that ultimately cause chronic inflammation.
The work by the team Nahrendorf and Swirski note, contributes to the knowledge regarding neutrophil activity, but there is still a lot of work to be done to learn more about how NETs form, or if their formation can be averted and of course, whether they behave the same way in humans.
More information: Neutrophil extracellular traps license macrophages for cytokine production in atherosclerosis Science 17 July 2015: Vol. 349 no. 6245 pp. 316-320. DOI: 10.1126/science.aaa8064
Abstract
Secretion of the cytokine interleukin-1β (IL-1β) by macrophages, a major driver of pathogenesis in atherosclerosis, requires two steps: Priming signals promote transcription of immature IL-1β, and then endogenous "danger" signals activate innate immune signaling complexes called inflammasomes to process IL-1β for secretion. Although cholesterol crystals are known to act as danger signals in atherosclerosis, what primes IL-1β transcription remains elusive. Using a murine model of atherosclerosis, we found that cholesterol crystals acted both as priming and danger signals for IL-1β production. Cholesterol crystals triggered neutrophils to release neutrophil extracellular traps (NETs). NETs primed macrophages for cytokine release, activating T helper 17 (TH17) cells that amplify immune cell recruitment in atherosclerotic plaques. Therefore, danger signals may drive sterile inflammation, such as that seen in atherosclerosis, through their interactions with neutrophils.
© 2015 Medical Xpress