Studies on peptide-modified cultures demonstrate control over both tissue growth and location in vitro
"In vitro fabricated biological tissue would be a valuable tool to screen newly synthesized drugs or understand the tissue development process," explain Takuya Matsumoto and his colleagues in a recent report. However tissues grown so far have not reached the size and final shape of natural tissue. Matsumoto and colleagues at Okayama, Osaka and Kansai Universities in Japan have now identified a tripeptide sequence that can be used to overcome both limitations. With hydrogel modified with the tripeptide they demonstrate enhanced tissue growth of a salivary gland both across a sheet of tissue and at specified localised points.
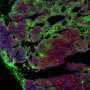
The submandibular gland develops a branched structure through budding and the elongation of ducts, which is also typical of other structures such as lungs, kidneys, mammary glands and spleen. Previous research has revealed the importance of the glycoprotein fibronectin in the morphological development and growth of salivary gland tissue. Consequently, Matsumoto and colleagues focused their investigations on the effects of the specific tripeptide sequence arginine-glycine aspartic acid (RGD) that acts as a cell binding site on fibronectin.
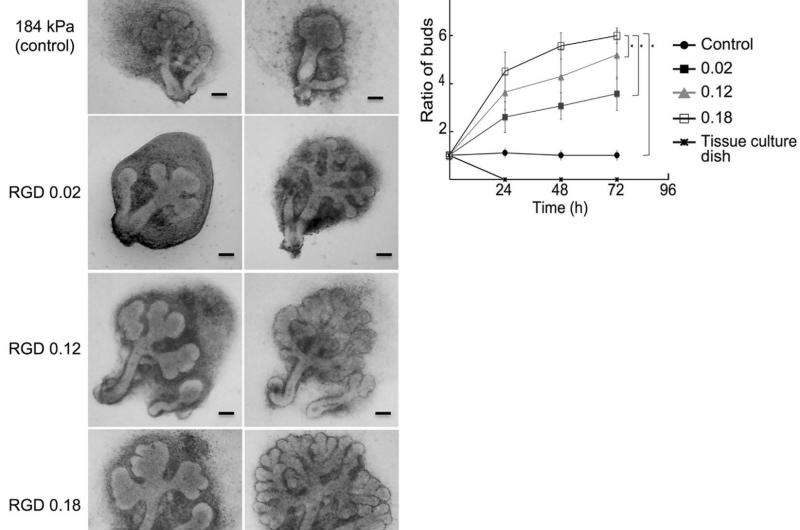
The researchers studied the growth of salivary gland tissue on hydrogel both modified with different concentrations of RGD and without and found significant growth enhancements that increased with RGD concentrations. They then experimented with RGD-modified hydrogel beads and demonstrated that localised growth enhancement occurred where the beads were situated, which could provide control over morphology in artificial tissue growth. Further studies also suggested that neuronal growth and the enhancement of growth factor proteins were responsible for the enhanced gland tissue growth observed.
In their report, the researchers conclude, "This in vitro SMG tissue growth modulation system can have a variety of uses including tissue arrays for drug screening and as a biologic tool to understand tissue development."
Tripeptide in previous studies
The shape development or morphogenesis of the submandibular gland develops through the formation of clefts that occur through a change in cell adhesion so that they bind to the surrounding extracellular matrix instead of each other. Previous research has shown that expression of the glycoprotein fibronectin is crucial for cleft formation.
Integrin is a receptor protein that bridges interactions between cells and the extracellular matrix. The domain of fibronectin that binds to integrin is the tripeptide sequence arginine-glycine aspartic acid (RGD) prompting studies of this tripeptide sequence on tissue growth.
Investigations have already shown that transplanting composites containing RGD peptides onto the back of a mouse can result in the development of growth plates – the tissue at the ends of bones in growing children and adolescents. However, to date, there have been no studies into the growth and morphology of tissues in vitro. The ability to grow tissues in vitro is particularly important for drug screening and studies to better understand tissue growth.
The role of FGFs
Fibroblast growth factors (FGFs) are a family of proteins involved in embryonic development, wound healing and signalling pathways. Two specific FGFs play a crucial role in the growth of the two main tissue types in the submandible salivary gland – FGF7 induces budding in the epithelial tissue and FGF10 induces the elongation of ducts in the mesenchymal tissue.
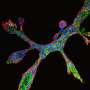
Matsumoto and colleagues imaged the salivary gland tissues grown in RGD-modified hydrogels using immunofluorescent staining and found both FGF7 and FGF10 were highly expressed. Experiments to grow the salivary gland tissues in the presence of FGF7 and FGF10 antibodies revealed that these antibodies inhibited tissue growth. The RGD-modified hydrogel was also ineffective at enhancing tissue budding in isolated epithelial cells alone.
The role of neurites
Previous studies have also suggested that neuron outgrowths – 'neurites' – from a region known as the parasympathetic ganglion in submandibular gland tissue are key to tissue growth. The researchers observed an even distribution of the neurite network throughout the gland tissues they had grown. Antibodies to the neurotrophic factors, which are responsible for neuron growth and survival, also inhibited submandibular gland tissue growth in RGD-modified hydrogels.
More information: "Peptide-modified Substrate for Modulating Gland Tissue Growth and Morphology In Vitro." Scientific Reports 5, Article number: 11468 (2015). DOI: 10.1038/srep11468