Extreme makeover of the heart: Matrix therapy is first FDA-approved procedure of its kind
A cardiovascular team at University of Utah Hospital has successfully performed a first-in-the-world heart procedure on a 72-year-old attorney after suffering a large heart attack. Amit N. Patel, M.D. M.S., director of clinical regenerative medicine and associate professor of surgery at University of Utah Health Care, was the first physician to perform an emerging heart procedure where cardiac matrix is directly injected into a damaged heart. This is the first clinical trial to be approved by the U.S. Food and Drug Administration (FDA) for matrix therapy of the heart.
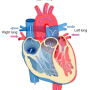
When an individual has a heart attack the heart tissue is damaged and the organ loses function. There is both scar tissue and weak heart muscle that remain, and eventually this leads to heart failure as tissue continues to die. Currently, there is no therapy to treat scar tissue in the heart.
Most approaches for regenerative therapy for the heart use stem cells or gene therapy. However, these therapies do not treat the scar tissue. The new procedure, endocardial matrix (ECM) is minimally invasive which allows doctors to do an extreme makeover of the heart from the inside out by injecting a natural matrix into damaged tissue while the patient is awake.
The procedure involves making a 3-D map of the patient's heart to see which part of the heart muscle is healthy, damaged and/or scarred. Once this is completed, the surgeon injects the matrix into the damaged and scarred tissue. The injection is made up of a mixture of VentriGelTM, a hydrogel derived from scaffolding protein that is extracted naturally from the heart.
"The new procedure involving ECM therapy is Cardiac Regeneration 2.0. It allows us to treat patients before they progress to heart failure, " said Patel. The patients will undergo MRIs of their heart at three-month intervals to objectively see if the ECM therapy resulted in an extreme makeover of the heart.
According to University medical staff, the first patient to receive the new treatment is back to her active lifestyle and working as an attorney in Northern Utah.
The FDA trial will enroll patients who have experienced a first heart attack within the past three years and have evidence of weak heart muscle or are at risk of heart failure. The FDA approved 18 trial spots for the first phase of the trial. The clinical trials will test the safety and efficacy of the procedure. It is expected that it will take at least three years until the procedure is made available to the public.
Approximately 785,000 new heart attack cases occur each year in the United States. Many of those patients will develop left ventricular dysfunction, a weakening of the heart's ability to effectively pump blood. According to the American Heart Association, at least 22 percent of men and 46 percent of women will develop heart failure—a debilitating and burdensome condition—within six years of their first heart attack.