Researchers compare direct gene vs. blood cell-mediated therapy of spinal cord injury
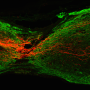
Spinal cord injury (SCI) leads to complex pathological changes that include the death of neurons and glial cells and degeneration of nerve fibers.
One promising approach for preventing neurodegeneration involves locally treating the site of injury. One such approach particularly suitable for SCI treatment is glial cell-derived neurotrophic factor (GDNF).
Previous studies of KFU researchers on direct gene therapy for spinal cord injuries indicated that the treatment resulted in the preservation of nervous tissue and functional recovery. Similarly, cell-based gene delivery has been shown to promote the growth of motor axons after partial and complete spinal cord transections.
The group of scientists from the Institute of Fundamental Medicine and Biology at Kazan Federal University developed special umbilical cord blood mononuclear cells, the main positive action of which was that they facilitated targeted delivery of the therapeutic molecules to motor neurons and thereby prolonged their survival. UCB-MCs have been used for gene delivery because of their suitability for low immunogenicity, accessibility and ease of production and storage, and because of the lack of legal, ethical and religious concerns related to using these cells.
A rat SCI model was used to examine the efficacy of the two methods.
At the 30th postoperative day after conducting the BBB (Basso, Beattie and Bresnahan) behavioral test on the animals and morphometric studies on their spinal cords, equal positive locomotor recovery in rats was observed after both direct and cell-based GDNF therapy.
Compared with direct gene injection, cell-mediated GDNF gene delivery led to considerably more pronounced preservation of myelinated fibers in the remote segments of the spinal cord (5 vs 3 mm from the epicenter), and this might depend on the expansion of the therapeutic influence in cell-mediated therapy over long distances as a result of the migration of the transplanted cells. UCB-MCs are suitable for cell therapy because they can potentially differentiate into not only blood cells, but also other types of cells such as myoblasts of heart and skeletal muscle tissues, hepatocytes, vascular endothelial cells, neurons, oligodendrocytes and astrocytes.
Transplantation of UCB-MCs is a promising strategy for enhancing posttraumatic spinal cord regeneration. Delivery of the GDNF gene into the site of injury holds considerable potential as a therapeutic intervention in SCI.
More information: Y O Mukhamedshina et al. "Adenoviral vector carrying glial cell-derived neurotrophic factor for direct gene therapy in comparison with human umbilical cord blood cell-mediated therapy of spinal cord injury in rat," Spinal Cord (2015). DOI: 10.1038/sc.2015.161