October 14, 2015 report
A new way to test for rejection or infection following lung transplantation
(Medical Xpress)—A team of researchers with Stanford University has found a new way to test for infection or impending rejection of lungs transplanted into a patient. In their paper published in Proceedings of the National Academy of Sciences, the team explains how their technique works and why it is so much safer than current methods.
As the researchers note in their paper, patients who undergo a lung transplant are far more likely to reject it then for any other organ. Making things even more difficult for doctors and their patients is that testing for infections is hit and miss, and testing for rejection involves cutting the patient open again and taking a biopsy, which puts the patient at additional risk of infection. In this new effort, the researchers report on a new technique they have developed that allows testing for both situations in a non-invasive manner.
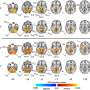
Human beings have some amount of freely flowing DNA bits in their blood all the time, they are known as circulating cell-free DNA, or just cfDNA. When an organ is taken out of one person and put into another, it carries with it the DNA of the donor, and some of it winds up in the recipient's bloodstream as cfDNA as well. In this new effort, the researchers performed sequencing on blood samples taken from 51 patients that had undergone lung transplants, both before and after the procedure. That allowed them to measure the amount of donor cfDNA in the patient that had received the organ. Analysis showed that those patients that rejected the lungs had a higher percentage of donor cfDNA in their blood. Thus, to predict imminent rejection, all the researchers had to do was take a blood sample and sequence it to measure the levels of cfDNA in their blood.
Next, the researchers wondered if it might be possible to use the same type of sequencing to determine if a patient was suffering from one of the common types of infections that occur after lung transplantation. They found that sequences of a common virus showed up in cfDNA that were consistent with clinical results, suggesting that such testing could be used to help identify patients during the early stage of infection.
More information: Iwijn De Vlaminck et al. Noninvasive monitoring of infection and rejection after lung transplantation, Proceedings of the National Academy of Sciences (2015). DOI: 10.1073/pnas.1517494112
Abstract
The survival rate following lung transplantation is among the lowest of all solid-organ transplants, and current diagnostic tests often fail to distinguish between infection and rejection, the two primary posttransplant clinical complications. We describe a diagnostic assay that simultaneously monitors for rejection and infection in lung transplant recipients by sequencing of cell-free DNA (cfDNA) in plasma. We determined that the levels of donor-derived cfDNA directly correlate with the results of invasive tests of rejection (area under the curve 0.9). We also analyzed the nonhuman cfDNA as a hypothesis-free approach to test for infections. Cytomegalovirus is most frequently assayed clinically, and the levels of CMV-derived sequences in cfDNA are consistent with clinical results. We furthermore show that hypothesis-free monitoring for pathogens using cfDNA reveals undiagnosed cases of infection, and that certain infectious pathogens such as human herpesvirus (HHV) 6, HHV-7, and adenovirus, which are not often tested clinically, occur with high frequency in this cohort.
© 2015 Medical Xpress