Researchers closer to better treatment for leading cause of hospital-acquired diarrhea
Researchers at Vanderbilt University Medical Center have obtained the crystal structure of a toxin from the bacterium Clostridium difficile ("C. diff")—the leading cause of hospital-acquired diarrhea in the United States.
Reporting in this week's Nature Microbiology, they also found that zinc is required to unleash the toxin's damaging effects in the colon. The discoveries are aiding efforts to develop vaccines and other novel therapies to prevent the potentially fatal consequences of C. diff infection.
"This is basic science. I think it gives a framework for understanding how, once you do have an infection, the toxins are causing the disease," said senior author D. Borden Lacy, Ph.D., associate professor of Pathology, Microbiology and Immunology and of Biochemistry.
Like anthrax, diphtheria and botulism, C. diff infection is a toxin-mediated disease. The bacterium actually produces two similar toxins, toxin A and toxin B. But unlike the other infections, there is as yet no vaccine or other treatment that can effectively block C. diff toxins.
Meanwhile, C. diff has become a major public health menace. In 2011, the bacterium caused nearly half a million infections in the United States, and approximately 29,000 people died from intestinal complications, including a form of colitis, within a month of the initial diagnosis.
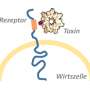
In 2012 and 2013, Lacy and her colleagues reported the mechanism by which toxin B kills cells. Earlier this year, they reported the identification of the cellular receptor that binds the toxin.
After binding to their receptors, the toxins are enveloped by an endosome, or tiny vesicle. Through a pore it drills into the cell membrane, each toxin then sends pieces of itself with two enzymatic activities into the cell. The enzymes modify the activity of cellular proteins, ultimately killing the cell.
In the current study, the researchers, led by Lacy's research assistant, Stacey Rutherford, generated the crystal structure of C. diff toxin A. Benjamin Spiller, Ph.D., associate professor of Pharmacology and of Pathology, Microbiology and Immunology, also contributed to the crystallography.
At Argonne National Laboratory outside Chicago, the researchers bounced a highly focused X-ray of a specific wavelength off the crystal. The resulting diffraction pattern was then converted using computational methods into a model of the toxin.
They found that one small section of the toxin is "highly conserved," meaning that its sequence of amino acids is identical to the same sequence in other Clostridium species.
This "suggests that antibodies specific for this conserved region could provide protection against multiple toxin-mediated clostridium infections and points to a generalizable strategy for generating safe vaccine antigens for this class of toxins," they concluded.
In addition, Nicole Chumbler, a graduate student in the Lacy lab who is now a postdoctoral fellow at Harvard Medical School, found that zinc is bound to the toxin and is required for its activity. Small molecules targeting the zinc-binding enzyme could block the toxin's effects.
Much remains to be discovered, Lacy said, but each study moves the dial a little closer to better treatment and prevention of C. diff infections.
More information: Nicole M. Chumbler et al. Crystal structure of Clostridium difficile toxin A, Nature Microbiology (2016). DOI: 10.1038/nmicrobiol.2015.2