Insulin-like growth factor linked to hippocampal hyperactivity in Alzheimer's disease
The mechanisms underlying the stability and plasticity of neural circuits in the hippocampus, the part of the brain responsible for spatial memory and the memory of everyday facts and events, has been a major focus of study in the field of neuroscience. Understanding precisely how a "healthy" brain stores and processes information is crucial to preventing and reversing the memory failures associated with Alzheimer's disease (AD), the most common form of late-life dementia.
Hyperactivity of the hippocampus is known to be associated with conditions that confer risk for AD, including amnestic mild cognitive impairment. A new Tel Aviv University study finds that the insulin-like growth factor 1 receptor (IGF-1R), the "master" lifespan regulator, plays a vital role in directly regulating the transfer and processing of information in hippocampal neural circuits. The research reveals IGF-1R as a differential regulator of two different modes of transmission—spontaneous and evoked—in hippocampal circuits of the brain. The researchers hope their findings can be used to indicate a new direction for therapy used to treat patients in the early stages of Alzheimer's disease.
The study was led by Dr. Inna Slutsky of TAU's Sagol School of Neuroscience and Sackler School of Medicine and conducted by doctoral student Neta Gazit. It was recently published in the journal Neuron. "People who are at risk for AD show hyperactivity of the hippocampus, and our results suggest that IGF-1R activity may be an important contributor to this abnormality," Dr. Slutsky concluded.
Resolving a controversy
"We know that IGF-1R signaling controls growth, development and lifespan, but its role in AD has remained controversial," said Dr. Slutsky. "To resolve this controversy, we had to understand how IGF-1R functions physiologically in synaptic transfer and plasticity."
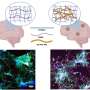
Using brain cultures and slices, the researchers developed an integrated approach characterizing the brain system on different scales—from the level of protein interactions to the level of single synapses, neuronal connections and the entire hippocampal network. The team sought to address two important questions: whether IGF-1Rs are active in synapses and transduce signalling at rest, and how they affect synaptic function.
"We used fluorescence resonance energy transfer (FRET) to estimate the receptor activation at the single-synapse level," said Dr. Slutsky. "We found IGF-1Rs to be fully activated under resting conditions, modulating release of neurotransmitters from synapses."
While acute application of IGF-1 hormone was found to be ineffective, the introduction of various IGF-1R blockers produced robust dual effects—namely, the inhibition of a neurotransmitter release evoked by spikes, electrical pulses in the brain, while enhancement of spontaneous neurotransmitter release.
A test for Alzheimer's?
"When we modified the level of IGF-1R expression, synaptic transmission and plasticity were altered at hippocampal synapses, and an increase in the IGF-1R expression caused an augmented release of glutamate, enhancing the activity of hippocampal neurons," said Gazit.
"We suggest that IGF-1R small inhibitors, which are currently under development for cancer, be tested for reduction aberrant brain activity at early stages of Alzheimer's disease," said Dr. Slutsky.
The researchers are currently planning to study how IGF-1R signaling controls the stability of neural circuits over an extended timescale.