ROBO1 helps cells put up stiff resistance
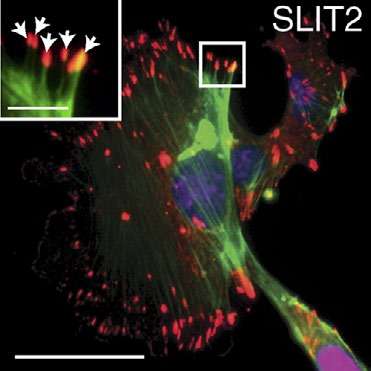
A protein called ROBO1 may delay the progression of breast cancer, according to a paper published in The Journal of Cell Biology. The study, "Loss of miR-203 regulates cell shape and matrix adhesion through ROBO1/Rac/FAK in response to stiffness" by Lily Thao-Nhi Le and colleagues, identifies a signaling pathway that may protect breast cells from the tumorigenic effects of stiff extracellular matrices.
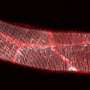
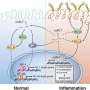
In living tissues, cells are surrounded by a matrix of secreted molecules. Changes in this extracellular matrix can stiffen tissues and activate oncogenic signaling pathways, but cells may try to protect themselves by reorganizing their cytoskeleton and remodeling their attachments to the matrix. The ROBO family of signaling proteins - best known for their role in guiding the growth of developing neurons - regulates the cytoskeleton, and may therefore help cells sense and respond to such changes in their environment.
A team of researchers led by Professor Lindsay Hinck at the University of California, Santa Cruz, examined the ROBO signaling pathway in breast epithelial cells and found that one family member, ROBO1, enhances cellular contractility and stimulates assembly of cell-matrix adhesions. Stiffer environments caused breast cells to downregulate a microRNA, miR-203, that normally suppresses Robo1, thereby elevating ROBO1 protein levels. This, in turn, enhanced cellular contractility and adhesion, allowing cells to retain their shape and position within stiff extracellular matrices.
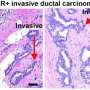
Breast cancer cells lacking ROBO1 were more invasive, suggesting that the upregulation of ROBO1 in stiff environments may prevent cells from metastasizing to other tissues. Moreover, ROBO1 has previously been shown to suppress cell proliferation, suggesting that this pathway could delay tumor progression. Accordingly, breast cancer patients whose tumors displayed low miR-203/high Robo1 expression had better long-term survival rates. Hinck and colleagues now want to confirm that ROBO1 can counteract the pro-tumorigenic effects of tissue stiffening in both mice and humans.
More information: Le, L.T., et al. 2016. J Cell Biol. dx.doi.org/10.1083/jcb.201507054