Bariatric embolization studied as potential treatment for morbid obesity
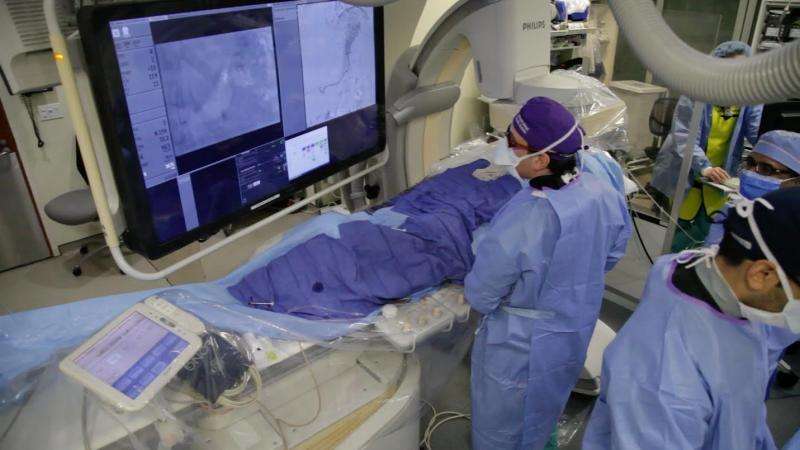
A multi-site clinical trial has been launched to assess the safety and effectiveness of bariatric arterial embolization, a minimally invasive procedure that can reduce the sensation of hunger, for treating patients with morbid obesity. The study is a research collaboration between the Icahn School of Medicine at Mount Sinai and Johns Hopkins University School of Medicine.
The Bariatric Embolization of Arteries for the Treatment of Obesity, known as the BEAT Obesity clinical trial, is studying how a minimally invasive procedure that targets ghrelin - the "hunger hormone" - affects weight loss in patients.
"The study is designed to evaluate if we can precisely modify the patient's sensation of hunger. The concept behind this study is that if we can reduce the amount of ghrelin circulating in the blood, patients will become less hungry," says Aaron Fischman, MD, Assistant Professor of Radiology and Surgery and Program Director of the Vascular and Interventional Radiology training program at the Icahn School of Medicine at Mount Sinai. "Ultimately, this may be an adjunct to bariatric surgery and one facet in a multidisciplinary approach to treating obesity."
More than two-thirds of adults are considered overweight or obese, according to the U.S. Department of Health and Human Services. Obesity has been linked to chronic health conditions that include hypertension, diabetes, heart disease, stroke, and cancer. Treatment can entail diet, exercise, medication and/or surgery.
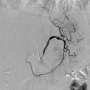
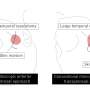
Investigators at Johns Hopkins developed a technique to target the cells that produce ghrelin by using embolization, a minimally invasive technique to deliver a material to block blood flow to those cells. Dr. Fischman has collaborated with Johns Hopkins investigators to develop a new method of delivery - the transradial approach, which is a less invasive way to enter the arterial system. Fischman pioneered the transradial technique in Interventional Radiology in 2012. He has implemented this method in the BEAT Obesity trial at Mount Sinai, where he is the lead site investigator. The procedure entails inserting a catheter through an artery in the wrist and directing it to vessels in the fundus portion of the stomach, where ghrelin is produced.
"There's less risk of bleeding, less pain and less trauma to the blood vessel compared to the traditional method which uses the femoral artery in the groin to access the arterial system. In addition, patients can ambulate immediately after the procedure, recover faster and more comfortably," says Fischman.
The image-guided procedure involves the injection of microscopic beads made of gelatin, which clog portions of the left gastric artery and cut off the blood supply.
Estimated enrollment for the clinical trial is 20. Participants have a body mass index (BMI) ranging from 40 to 50, which is above the obesity threshold level of 30. Preliminary results show a decrease in appetite corresponding to lower levels of circulating blood levels of the hunger hormone. Patients have shown promising weight loss in the first month after the procedure and an average excess weight loss of roughly 10 percent at 3 months.
Further research is needed to study the effects of the procedure over longer periods of time.
The principal investigator for this study is Aravind Arepally, MD, Piedmont Healthcare. Co-investigators include: Clifford Weiss, MD, and Dara Kraitchman, VMD, PhD, at Johns Hopkins and Rahul Patel, MD, Mount Sinai.
This study is expected to be completed in June 2016. The National Institute of Biomedical Imaging and Bioengineering and Merit Medical, Inc. supported this research.