Drug shows promise against rare, aggressive skin cancer
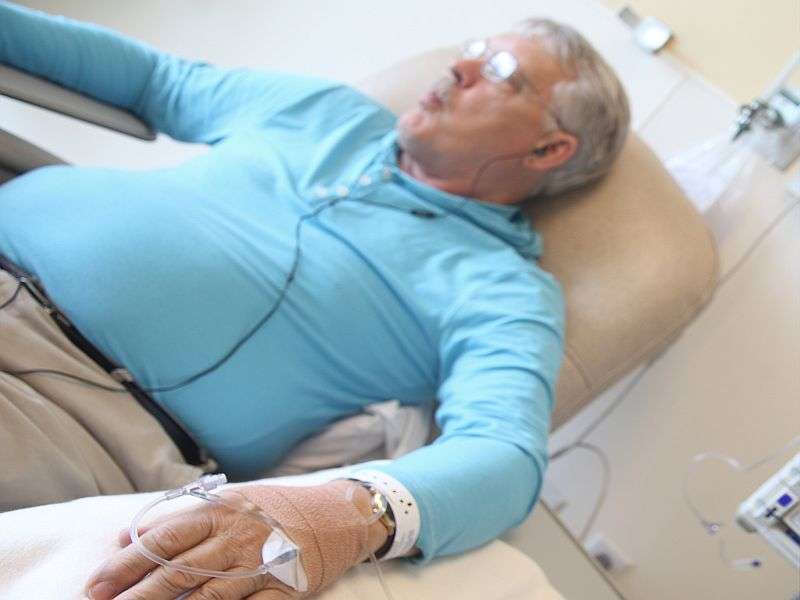
(HealthDay)—A newer drug that boosts the immune system's ability to kill tumor cells may help people with a rare, aggressive form of skin cancer, a preliminary study suggests.
The intravenous drug, marketed as Keytruda, is already used to treat some advanced cases of melanoma, another dangerous form of skin cancer. The new study tested it against a skin tumor called Merkel cell carcinoma (MCC).
Most people have probably never heard of the cancer, but MCC is deadlier than melanoma, said Dr. Paul Nghiem, a professor of medicine at the University of Washington, who led the new study.
When the disease reaches an advanced stage, chemotherapy is an option—but not a good one, Nghiem said.
"Chemotherapy will often shrink the cancer," he said. "But it comes back quickly, and even angrier."
Plus, chemo can take a toll on the immune system. "And that's a very bad idea in these patients," Nghiem said.
Keytruda (pembrolizumab) is one of a new class of drugs that block a "pathway" called PD-1. That frees up the immune system to attack cancer cells. In the United States, the drug is approved for treating certain cases of lung cancer and advanced melanoma that no longer respond to other drugs.
In the new study, Nghiem's team gave the drug to 26 patients with advanced MCC. Most had metastatic cancer, meaning it had spread beyond lymph nodes near the original skin tumor.
Overall, out of 25 people who were evaluated, 14 patients—or 56 percent—saw their cancer shrink at least partially. In four patients, all signs of the cancer disappeared.
And after more than six months of follow-up, the cancer remained under good control in 12 of the 14, Nghiem said.
The findings were scheduled for release Tuesday at the annual meeting of the American Association for Cancer Research in New Orleans, and published online in the New England Journal of Medicine.
An oncologist who was not involved in the study called the results "impressive."
"For these patients, the response to chemotherapy typically lasts two to four months at best," said Dr. Nikhil Khushalani, of the Moffitt Cancer Center in Tampa, Fla. "And metastatic MCC is invariably fatal."
Given the results so far, Khushalani said, it's "likely" the treatment could extend patients' lives.
Each year, about 1,500 Americans are diagnosed with MCC, according to the American Cancer Society. It mainly strikes older adults, and sometimes people with a compromised immune system—such as organ transplant patients and people with HIV.
But most people who develop MCC do not have a suppressed immune system, Nghiem said.
In recent years, researchers have discovered a virus, dubbed Merkel cell polyomavirus, which seems to contribute to many cases of MCC.
Researchers believe that most people are infected with the virus, but the immune system keeps it in check. It's not clear why or how it feeds MCC development.
Other cases of MCC are tied to excessive ultraviolet exposure from the sun, Nghiem said.
Of the 26 patients in this study, 17 had tumors that carried the Merkel polyomavirus. All patients received Keytruda every three weeks—for between four and 49 weeks.
A higher percentage of patients with virus-positive tumors responded to the drug. But Nghiem said the difference was not significant in statistical terms, so the treatment seems to work whether the virus is present or not.
There were side effects, with fatigue a common one, Nghiem said.
In general, a danger with Keytruda is that it can damage healthy body tissue. In this study, two patients developed signs of inflammation in the liver or heart muscle and had to discontinue the drug after only one or two doses.
Yet, both patients still showed a response to the drug months after stopping it, Nghiem said.
That touches on a key question: How long do patients need to stay on the drug? One reason that's important, Nghiem noted, is cost. The drug's manufacturer, Merck, priced it at about $12,500 per month.
Nghiem said he suspects patients will vary in how long they need to take the drug.
Keytruda is not yet approved for treating MCC. One way for patients to get the drug would be to enroll in a clinical trial like the current one, Nghiem said.
Merck and the U.S. National Cancer Institute are funding Nghiem's trial.
More information: The American Cancer Society has more on Merkel cell carcinoma.
Copyright © 2016 HealthDay. All rights reserved.