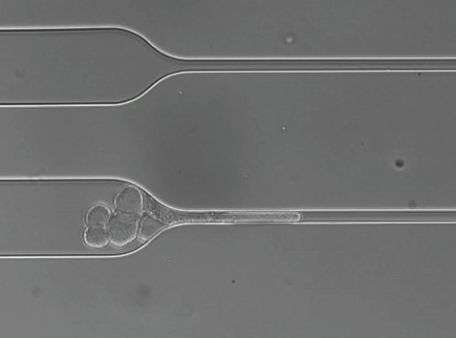
Metastasis-promoting circulating tumor cell clusters pass through capillary-sized vessels
Massachusetts General Hospital (MGH) investigators have found that circulating tumor cell (CTC) clusters—which are more efficient in spreading cancer throughout the body than are single CTCs—can pass through capillary-sized blood vessels. Their findings, which contradict the widely-held belief that CTC clusters are too large to pass through capillaries, are being published online in Proceedings of the National Academy of Sciences and suggest potential strategies to reduce clusters' metastatic potential.
"By showing that aggregates containing dozens of cancer cells can unfold to pass through capillary-sized constrictions, we've challenged the current viewpoint that distant organ metastases are 'seeded' only by individual CTCs," says Sam Au, PhD, of the MGH Center for Engineering in Medicine, lead author of the report. "This information may change the standard narrative of how metastasis initiates and allow us to devise better ways to combat it."
The enhanced ability of CTC clusters to drive metastasis has been recognized for decades and supported by recent studies from the MGH Cancer Center and elsewhere. But details of the clusters' behavior within the circulation have not been explored, and the fact that even large clusters have been found in the veins of patients' arms, far from the location of primary tumors, suggested to the investigators that clusters must be able to pass through capillaries.
To investigate this hypothesis, they developed a microfluidic device with channels that taper to widths of 5 to 10 micrometers (millionths of a meter), the same as the smallest human blood vessels. In a series of experiments using CTC clusters isolated from patient blood samples, from cancer cell lines and from cultured clusters, they found that clusters of 20 cells or more were capable of passing through even the smallest constrictions in the device.
Detailed imaging revealed that, upon encountering a constriction, CTC clusters unfold into a series of single cells, like beads on a chain, and pass through in single file. On the other side of the constriction, the cells spontaneously refold into a cluster, which appears to be undamaged and retains its ability to proliferate. Within constrictions, cells stay connected to each other by means of pre-existing cell-to-cell interactions within clusters, with the strongest contacts probably being maintained while the weaker connections are temporarily lost.
The speed with which clusters pass through a constriction depends on the size of the largest cell, not the overall size of the cluster. Although treating samples of CTC clusters from patients with factors known to disrupt cellular adhesion, including the chemotherapy drug paclitaxel, does not immediately change the form or viability of clusters, the treated clusters do not reassemble after passing through a constriction, and the cellular chains break apart into single cells or smaller aggregates.
To examine the behavior of CTC clusters in the blood vessels of live animals, the researchers used embryonic zebrafish, which have major blood vessels the size of human capillaries and are transparent, making it easy to observe the passage of labeled CTC clusters. Just as within the microfluidic constriction device, human CTC clusters pass through larger and smaller vessels in the same sort of single-file configuration as within the device and then reassemble.
"Since how clusters behave at constrictions depends on how strongly the cells adhere to each other, if we can change that strength - either by breaking clusters up into individuals cells or preventing them from unfolding, we might be able to control their ability to pass through narrow vessels," says Au, who is a research fellow in the MGH Department of Surgery.
Mehmet Toner, PhD, director of the BioMicroElectroMechanical Systems Resource Center in the MGH Department of Surgery and senior author of the PNAS report, adds, "Among the things we need to investigate now are which adhesion molecules are most important to this process, exactly how clusters reform themselves - either repairing broken adhesions or forming new ones - after exiting constrictions, and whether the forces exerted on clusters as they pass through capillaries contribute to their metastatic potential. If we can develop ways of impeding that passage, we might be able to reduce the chance of metastasis, which is the leading cause of cancer death."
More information: Clusters of circulating tumor cells traverse capillary-sized vessels, PNAS, www.pnas.org/cgi/doi/10.1073/pnas.1524448113