Scientists discover genetic basis of reactive oxygen species increase
An international team of scientists has determined under which conditions a body produces more superoxide—a dangerous form of oxygen able to destroy DNA. The article was published in Free Radical Biology and Medicine.
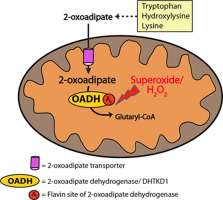
Human mutations in a gene encoding the DHTKD1 protein result in a range of neurological disturbances. On a molecular level, the mutations cause accumulation of the reactive oxygen species (ROS) and degradation products of lysine and tryptophan. The structure of the DHTKD1 protein is similar to an enzyme, 2-oxoglutarate dehydrogenase, which can produce superoxide—a dangerous form of oxygen able to destroy cellular components including DNA. An international scientific team including Victoria Bunik, a leading researcher of the Lomonosov Moscow State University, showed under which conditions the DHTKD1 protein induces production of the superoxide. According to the researcher, further study of DHTKD1 could lead to methods for curing the mutations in a gene encoding this protein.
A cellular nuclear power plant
Mitochondria are often compared to an NPP inside a cell. Its fuel consists of fatty acids and pyruvate, which finally produce ATP—an all-purpose source of energy for in-cell processes. Like nuclear power plants, mitochondria are complex in operation, interact with dangerous substances and produce waste in the form of free radicals (chemical compounds containing one or several unpaired electrons in the outer shell, which makes them negatively charged and highly active. Here is found the superoxide—a toxic type of oxygen that can harm cellular components including DNA, thus impairing cellular homeostasis. A cell combats the toxic forms of oxygen using antioxidants and antioxidant defense system proteins, for example, superoxide dismutase.
The adipate twins
Disturbed metabolism of amino acids may reduce synthesis of enzymes, proteins catalysing particular chemical reactions in the body. So a shortage of the amino acids lysine and tryptophan may cause physical, neurologic and mental malfunctions, and even lead to death. Those amino acids cannot be produced from other substances in human body and must come from food. 2-aminoadipate and 2-oxoadipate are the degradation products of lysine, tryptophan and hydroxylysine.
Recently, scientists found a connection between aciduria (increased acidity of urine, showing serious malfunction in metabolism) in patients with raised content of 2-aminoadipate and 2-oxoadipate and gene mutations in the DHTKD1 protein. That confirms an earlier hypothesis of the MSU researchers (Bunik & Degtyarev, 2008) that an enzyme encoded by DHTKD1 oxidizes 2-oxoadipate.
The DHTKD1 protein is produced in higher quantities in liver and kidney cells, where lysine and tryptophan are more actively degraded, and in human skeletal muscles. Both the down- and up-regulation of the DHTKD1 expression increase the level of reactive, dangerous forms of oxygen.
the Lomonosov Moscow State University scientist Victoria Bunik was invited to participate in the research as an international expert in the multienzyme complexes of 2-oxo acid dehydrogenases, which also include the above mentioned 2-oxoglutarate dehydrogenase, similar to the DHTKD1 protein, and their side-reactions of ROS formation. In 2003, Victoria Bunik published a review of her own studies on the mechanism of such side reactions, and in 2008, published a work predicting the DHTKD1-dependent expression of the 2-oxoadipate dehydrogenase previously unknown in mammals.
The current publication on the DHTKD1 enzyme was dedicated to experimental research on a generation of active oxygen forms, catalyzed by the 2-oxoadipate dehydrogenase complex. In this study, biochemical methods were used to register the amount of hydrogen peroxide produced by mitochondria. A specific fluorescent compound was created for that purpose. The researchers observed that the production of the superoxide increases under high concentrations of 2-oxoadipate, and also detected that it is exactly the 2-oxoadipate oxidization that causes the formation of the superoxide.
"Apart from characterization of a new potential source generating ROS, the scientific significance of the study demonstrates a high level of understanding of living systems attained in fundamental research. This allowed us not only to decipher the genome-coded information in terms of the DHTKD1 function, but also to predict the behavior of a biological system (i.e. mitochondria) under variety of conditions," says Victoria Bunik. "The research promotes developing therapies to cure such patients," the scientist concludes.