Halting protein degradation may contribute to new cancer treatment
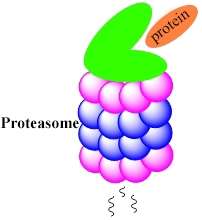
De Bruin carries out chemical-biological research on proteasomes. 'They are a kind of degradation factory that cuts proteins in cells into smaller fragments. It's a highly useful activity: it's a way for cells to clean up proteins that are redundant or that have been damaged.'
Proteasome inhibitors against cancer
But there are times when it can be useful to prevent proteasome activity, with auto-immune diseases and cancer, for example. De Bruin explains, 'With particular types of cancer, protein production in the cell is much higher than normal. If you inhibit the proteasomes, this stops degradation and the proteins pile up, which eventually kills the cancer cell.' There are already proteasome inhibitors on the market for treating Kahler's disease (multiple myeloma), for example. The disadvantage of these inhibitors is that they affect proteasome activitity not only in cancer cells, but also in healthy cells, which means they have many side-effects.
Chemical measuring method
De Bruin studies methods of inhibiting proteasome activity more specifically. He has designed a method to measure all the activities of proteasomes at the same time. Proteasomes are cylindrical in shape; the proteins to be broken down are funnelled through the cylinder where they are cut through at different places. Proteasomes have six different active centres, that act as 'scissors' that selectively cut the proteins. De Bruin: 'I have made small molecules that can bind to these active centres. We can now study the activity of all six active centres at the same time.'
Specific inhibitors
He then designed molecules that can also bind to the active centres in the proteasomes, but the aim in this case is to halt their activity. He produced one specific inhibitor for each active centre. These inhibitors can now be used to study the role that each active centre plays in different diseases, such as cancer and auto-immune conditions. The experiments showed it was possible to kill leukaemia cells by selectively blocking two of the six active centres.
Promising
De Bruin has published his findings in scientific articles and his 'tools' are now in wide use. There is still a lot to learn about how these proteasomes work precisely and about their role in cancer and auto-immune diseases; biologists can gain new insights by carefully measuring their activity and halting the active centres. De Bruin believes that selectively inhibiting the different proteasome centres holds promise for the future. 'These kinds of inhibitors may have fewer side-effects than the medicines that are currently in use. But the research still has a long way to go.'