Sticky molecules to tackle obesity and diabetes
Okayama University researchers report that the over-expression of an adhesion molecule found on the surface of fat cells appears to protect mice from developing obesity and diabetes. The findings, published in the journal Diabetes, March 2016, may fuel the development of new therapies targeting these diseases.
The molecular mechanisms that trigger the development of obesity and diabetes are still the subject of ongoing research. For example, limited information exists regarding the role of adhesion molecules in fat tissues – cell surface proteins that help cells stick to their surroundings or to each other. Scientists believe adhesion molecules may affect the build-up of excess fat during obesity. One such molecule, ACAM (adipocyte adhesion molecule), has been found on the surface of fat cells (or adipocytes) in rats, mice and humans, but its functional role remains unclear.
Now, Jun Wada and co-workers at Okayama University, Japan, have shown that transgenic mice engineered to express high levels of ACAM appear to be protected from developing obesity and diabetes.
Cellular biology and obesity
Previous studies have shown that, when rats are fed a high-fat diet, the extracellular matrix (ECM) surrounding mature adipocytes undergoes remodelling. As the ECM is an important regulator of adipocyte functioning, it follows that this remodelling would have knock-on effects on the biology of the fat cells. The adhesion molecules on the adipocyte cell surfaces play a key role in interacting with, and binding to, the ECM, but little obesity-related research has focused on these crucial components, until now.
Wada and his team generated three lines of transgenic mice, with high, intermediate and low levels of ACAM, triggered by aP2 promoter. The groups of mice were fed on a high fat, high sugar diet of chow, and the researchers discovered that the group with high ACAM expression were protected from obesity and diabetes, despite their diet.
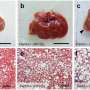
Compared with wild-type mice used as a control group, the body fat of the aP2-driven ACAM mice was significantly lower, and adipocyte size was considerably reduced.
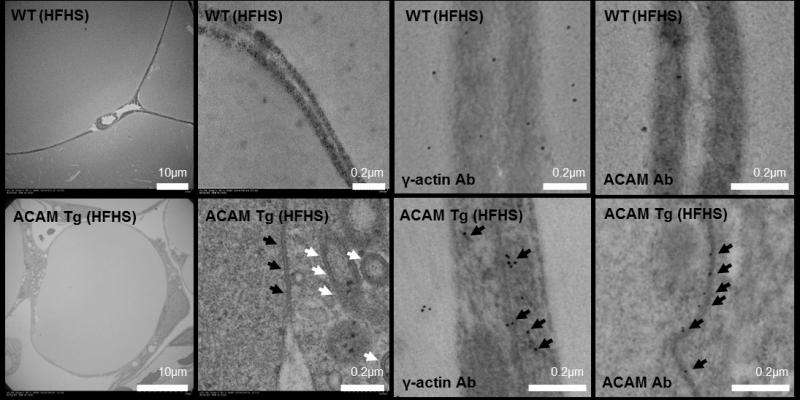
ACAM was stained to show up on scans, so that the team could track its behavior in the body. They found that ACAM was expressed abundantly on adipocyte cell plasma membranes, and influenced the development of zonula adherens structures via γ-actin polymerization. The development of zonula adherens in turn may enhance the mechanical strength of the fat cell surface, reducing its ability to grow larger and increase in volume. The process also appears to have a knock-on effect improving insulin sensitivity and glucose tolerance by influencing signalling processes.
Targeting ACAM in this way could provide a candidate for new therapies, though extensive further research is required.
The researchers generated mouse lines capable of higher levels of ACAM expression than wild-type mice. They engineered three groups of mice to express varying levels of ACAM expression driven varying promoter activities of 'aP2' – an adipocyte fatty acid binding prptein. Fluorescent tagging also allowed the team to monitor how ACAM behaved, and how it influenced other molecules inside the body.
Wada's team fed the mice on a high fat, high sugar diet. They found that, in the group with the highest levels of ACAM, the mice appeared to be completely protected from obesity and diabetes. ACAM was abundantly expressed on mature adipocyte plasma membranes, and interacted with two further molecules, myosin II-A and γ-actin. These interactions facilitated the formation of so-called 'zonula adheren' structures on the cell surface. The researchers believe the zonula adherens may increase the mechanical strength of the adipocyte cell surface, restricting increases in cell volume and the accumulation of fat cells. The resulting alterations in molecular signalling may also improve insulin sensitivity and glucose tolerance.
In their paper published in May 2016 in Diabetes, Wada's team hold great hopes for their results: "Our finding provides new therapeutic modalities targeting the processes of cell adhesion and actin polymerization of adipocytes in the treatment of obesity and diabetes."
More information: Kazutoshi Murakami et al. Antiobesity Action of ACAM by Modulating the Dynamics of Cell Adhesion and Actin Polymerization in Adipocytes, Diabetes (2016). DOI: 10.2337/db15-1304