Strengthening immune defence may be solution for treating tuberculosis
Researchers at Linköping University have made a discovery that could contribute to developing new vaccines and treatment alternatives for tuberculosis in the future. The results have been published in Scientific Reports, a sister journal to the highly respected periodical Nature.
Eight million people suffer from – and two million die from – tuberculosis every year. For HIV patients, tuberculosis is the leading cause of death. Antibiotics are currently used against tuberculosis, as no effective vaccine could be developed thus far – but with growing resistance to antibiotics, the cure for tuberculosis is threatened.
"When the world of research is looking for medicines, finding ways to strengthen immune defence is also now desirable, since medicines that attach the bacteria directly have been shown to lead to the development of resistance to antibiotics. Strengthening the immune defence system can be done through things like what is known as 'stimulated autophagy'. But our research shows that the most common signal path for stimulating autophagy can be immediately dangerous," says Robert Blomgran, research fellow in medicinal microbiology at Linköping University.
Autophagy is an important process that occurs naturally in the body to clean out defective proteins and components that could otherwise damage the cells and cause illness. Making use of this process – what we call 'stimulated autophagy' – could be one way of also protecting the body against intracellular microbes. Currently, the most common pharmacological activation of autophagy occurs through what is known as mTORC1 inhibition.
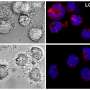
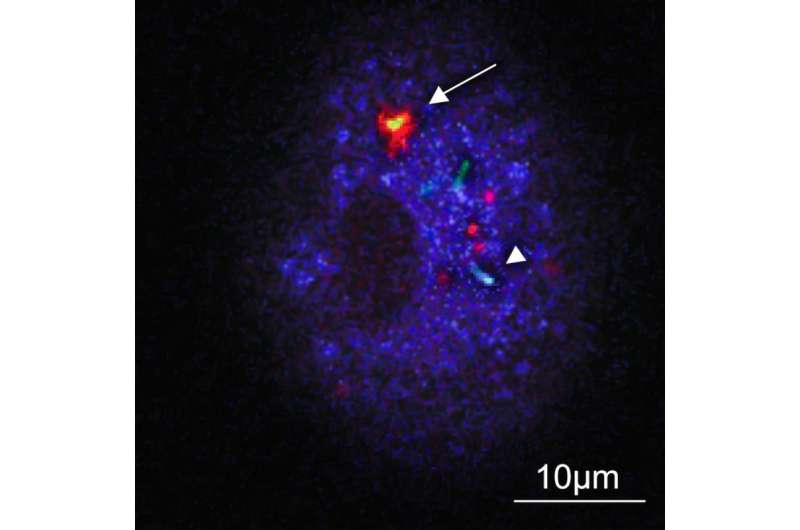
What the researchers discovered was that when the tubercule bacilli are few – which they are when the illness has not yet broken out – stimulated autophagy through mTORC1 inhibition is harmful. Instead, the process leads to the tubercule bacilli multiplying within the host cell, which in patients could involve the risk that the tuberculosis worsens.
If the TB patient also has HIV, that illness worsens. This is why the researchers looked more closely at what effects the induction of autophagy has in immune cells infected with both HIV and tuberculosis. The experiments showed that autophagy was then exceptionally dangerous, with an uncontrolled growth of tubercule bacilli as a result.
"How autophagy is stimulated needs to be reviewed so that over the long term it can have the desired effect in patients with tuberculosis, and in the extra sensitive patient group with both tuberculosis and HIV. If this is taken into consideration when new medicines are tested, setbacks could be prevented," Mr Blomgran says.
The research group will now be working further on research concerning other ways to stimulate autophagy and strengthen immune defence cells such as macrophages and dendritic cells, thus controlling the tubercule bacilli.
More information: Anna-Maria Andersson et al. Autophagy induction targeting mTORC1 enhances Mycobacterium tuberculosis replication in HIV co-infected human macrophages, Scientific Reports (2016). DOI: 10.1038/srep28171