Ouch! Flu spray fails again, panel urges shot instead
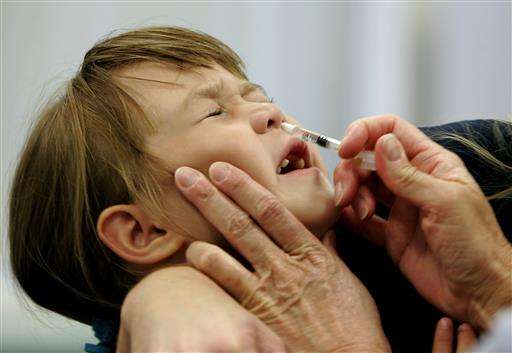
The nasal spray version of the annual flu vaccine failed to protect kids again last year, the latest in a string of failures that has prompted an expert panel to recommend that doctors stop giving it to patients.
Health officials reported Wednesday that the spray performed dismally for the third straight year, while the traditional flu shot—the one that stings—worked reasonably well this winter.
"We could find no evidence (the spray) was effective," said Dr. Joseph Bresee, a flu expert at the U.S. Centers for Disease Control and Prevention.
The product, AstraZeneca's FluMist, was once regarded as the best vaccine for protecting children against flu. Only two years ago, officials advised doctors that whenever possible they should use FluMist on young kids instead of traditional flu shots.
On Wednesday a federal advisory committee on immunization voted to retract its endorsement of the vaccine after preliminary CDC study results presented to the committee showed it provided no protection from the flu strain that made most people sick last year.
Recommendations from the committee are usually adopted by the government, which sends the guidance out to doctors.
Most Americans get the vaccine in the form of a shot.
FluMist is the only spray-in-the-nose vaccine on the market. It was first licensed in 2003 and is approved for healthy people ages 2 to 49. Unlike shots made from a killed virus, it is made from a live but weakened flu virus.
Early studies, including randomized trials coordinated by the manufacturer, showed FluMist was highly effective, and in children performed better than traditional flu shots.
The numbers looked so good that in 2014 the committee made the unusual decision to advise doctors that FluMist was better at preventing flu in healthy kids ages 2 through 8.
But some, including a representative of the American Academy of Pediatrics, opposed the move. They argued FluMist is more expensive, and that FluMist's reputation hinged on studies that were done before flu vaccine was encouraged for most children and when vaccination rates were much lower.
It's not clear why the vaccine isn't working. Bresee said some suspect that it is has to do with the decision a few years ago to incorporate four strains of flu in FluMist instead of the traditional three. It could be that the added strain reduced the body's response to another strain, he said.
An AstraZeneca spokesperson said the company is evaluating the committee's recommendation.
Many doctors, hospitals and flu clinics place their orders for flu vaccine early in the year. So the panel's decision may pose a problem for fall flu vaccination campaigns that target kids.
"It would really disrupt the vaccine supply," said Dr. William Schaffner, a Vanderbilt University vaccine expert.
More information: Panel: www.cdc.gov/vaccines/acip
© 2016 The Associated Press. All rights reserved.