Immune-based therapy in mice shows promise against pancreatic cancer
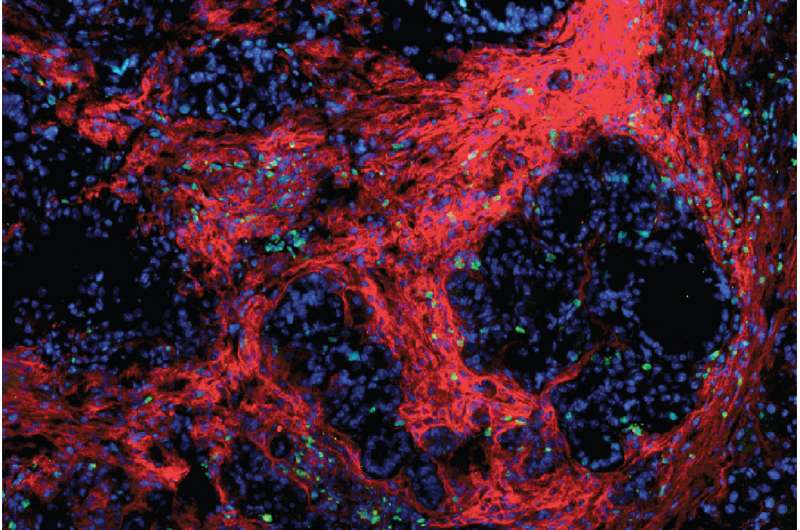
While immune therapy has proven effective in treating certain types of cancer, especially lung cancer and melanoma, tumors of the pancreas remain among the most difficult to treat and, so far, are impervious to immune-based therapies. Now, a new study in mice has shown that immunotherapy against pancreatic cancer can be effective when given in conjunction with drugs that break up the fibrous tissue in these tumors.
The study, from Washington University School of Medicine in St. Louis, appears July 4 in Nature Medicine.
Based on the study's findings, doctors at Siteman Cancer Center at Washington University School of Medicine and Barnes-Jewish Hospital are conducting a phase 1 clinical trial in patients with advanced pancreatic cancer that will test the safety of this drug combination when given along with standard chemotherapy.
"Pancreatic tumors are notoriously unresponsive to both conventional chemotherapy and newer forms of immunotherapeutics," said senior author David G. DeNardo, PhD, an assistant professor of medicine. "We suspect that the fibrous environment of the tumor that is typical of pancreatic cancer may be responsible for the poor response to immune therapies that have been effective in other types of cancer."
Unlike other cancer types, pancreatic tumors are characterized by a large amount of what DeNardo describes as scar tissue. This extra connective tissue and the cells that deposit it provide a protective environment for cancer cells, stopping the immune system from attacking the tumor cells and limiting the cancer's exposure to chemotherapy delivered through the bloodstream. DeNardo and his colleagues investigated whether some of this protection might be lost if they could disrupt the proteins that help fibrous tissue adhere to itself and surrounding cells.
"Proteins called focal adhesion kinases are known to be involved in the formation of fibrous tissue in many diseases, not just cancer," DeNardo said. "So we hypothesized that blocking this pathway might diminish fibrosis and immunosuppression in pancreatic cancer."
Focal adhesion kinase (FAK) inhibitors have been developed in other areas of cancer research, but DeNardo and his colleagues, including oncologist Andrea Wang-Gillam, MD, PhD, an associate professor of medicine, are the first to test them against pancreatic cancer in conjunction with immunotherapy. In the mouse study, an investigational FAK inhibitor was given in combination with a clinically approved immune therapy that activates the body's T cells and makes tumor cells more vulnerable to attack.
Mice with a model of pancreatic cancer survived no longer than two months when given either a FAK inhibitor or immune therapy alone. Adding FAK inhibitors to standard chemotherapy improved tumor response over chemotherapy alone. But the three-drug combination—FAK inhibitors, immune therapy and chemotherapy—showed the best outcomes in laboratory studies, more than tripling survival times in some mice. Some were still alive without evidence of progressing disease at six months and beyond.
The success of this mouse study provided a strong rationale for testing this drug combination in patients with advanced pancreatic cancer, according to Wang-Gillam.
"This trial is one of about a dozen we are conducting specifically for pancreatic cancer at Washington University," she said. "We hope to improve outcomes for these patients, especially since survival with metastatic pancreatic cancer is typically only six months to a year. The advantage of our three-pronged approach is that we are attacking the cancer in multiple ways, breaking up the fibers of the tumor microenvironment so that more immune cells and more of the chemotherapy drug can attack the tumor."
More information: Jiang H, Hegde S, Knolhoff BL, Zhu Y, Herndon JM, Meyer MA, Nywening TM, Hawkins WG, Shapiro IM, Weaver DT, Pachter JA, Wang-Gillam A, DeNardo DG. Targeting focal adhesion kinase renders pancreatic cancers responsive to checkpoint immunotherapy. Nature Medicine. July 4, 2016. DOI: 10.1038/nm.4123

















